PIPELINE
OVERVIEW
ABBV-319 is an investigational drug under clinical development. Safety and efficacy have not been established.
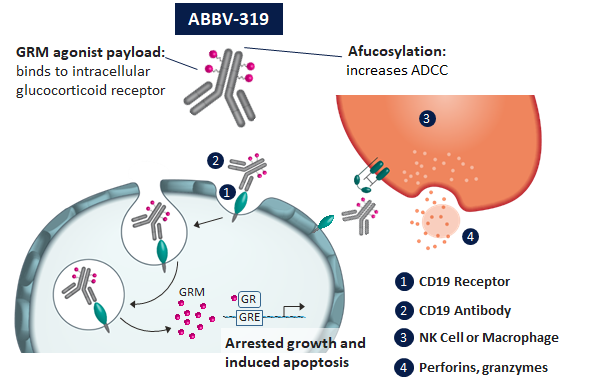
ABBV-319 is a CD19-GRM antibody-drug conjugate (ADC) composed of an optimized high-affinity immunoglobulin G1 antibody conjugated to a potent proprietary glucocorticosteroid payload.
PROPOSED MECHANISM OF ACTION
Systemic glucocorticosteroids (ie, glucocorticoid receptor modulators [GRMs]) show robust monotherapy activity against B-cell malignancies at high doses; however, they may be accompanied by steroid-associated toxicities that limit dosing.
CD19 is a marker essential for B-cell proliferation and has high expression across B-cell malignancies including diffuse large B-cell lymphoma (DLBCL), follicular lymphoma (FL), and chronic lymphocytic leukemia (CLL).
ABBV-319 is a CD19-GRM antibody-drug conjugate (ADC) composed of an optimized high-affinity immunoglobulin G1 antibody conjugated to a potent proprietary glucocorticosteroid payload. ABBV-319 is a multimodal ADC that takes advantage of both:
- Enhanced antibody-dependent cellular cytotoxicity (ADCC) via afucosylation
- Targeted GRM payload delivery to maximize anticancer activity while lowering the risk of systemic steroid toxicities
DEVELOPMENT
ABBV-319 is being investigated in a phase 1 trial for the treatment of B-cell malignancies, specifically DLBCL, FL and CLL.
CLINICAL TRIALS
View select clinical trials with ABBV-319 now. To view a full list of clinical trials in which ABBV-319 is being investigated, please visit ClinicalTrials.gov.
- Levy M, et al. A First-in-Human Phase 1 Study of ABBV-319, an Antibody-Drug Conjugate Composed of a CD19 Antibody Linked to a Potent Propriety Glucocorticosteroid, in Patients with Relapsed or Refractory B-Cell Malignancies. Poster. 64th ASH. December 10-13, 2022. New Orleans, LA.
OVERVIEW
ABBV-453 is an investigational drug under clinical development. Safety and efficacy have not been established.
ABBV-453 is an oral B-cell lymphoma 2 (BCL-2) inhibitor being investigated in relapsed or refractory (R/R) multiple myeloma (MM).
PROPOSED MECHANISM OF ACTION
B-cell lymphoma 2 (BCL-2) is an anti-apoptotic, pro-survival member of the BCL-2 family of proteins that function to regulate the intrinsic pathway of apoptosis, or programmed cell death, and help maintain cellular homeostasis.1,2 BCL-2 is a pro-survival oncoprotein which can be overexpressed as a result of cancer-specific mutations or gene amplifications.3
MM is a heterogenous disease with IgH translocations such as t(11;14), t(4;14), t(6;14), t(14;16), and t(14;20) in approximately 40% of MM patients. 4 The most common IgH translocation in MM is t(11;14). BCL-2 dependency varies across MM; MM cells with the t(11;14) translocation are primarily dependent on BCL-2 for survival. ABBV-453 binds to BCL-2, preventing it from binding to pro-apoptotic proteins and thereby restoring the cell's ability to undergo apoptosis.
DEVELOPMENT
ABBV-453 is being investigated in a Phase 1 trial for the treatment of R/R MM.
CLINICAL TRIALS
View select clinical trials with ABBV-453. To view a full list of clinical trials in which ABBV-453 is being investigated, please visit ClinicalTrials.gov.
References
- Cory S, et al. The Bcl-2 family: roles in cell survival and oncogenesis. Oncogene. 2003;22:8590-8607.
- Plati J, et al. Apoptotic cell signaling in cancer progression and therapy. Integr Biol (Camb). 2011;3:279-296.
- Davids MS, et al. Targeting the B-cell lymphoma/leukemia 2 family in cancer. J Clin Oncol. 2012;30(25):3127-3135.
- Rajkumar SV, et al. Multiple myeloma: 2022 update on diagnosis, risk stratification, and management. Am J Hematol. 2022;97:1086-1107.
- Touzeau C, et al. Targeting Bcl-2 for the treatment of multiple myeloma. Leukemia. 2018;32:1899–1907.
OVERVIEW
CLBR001+SWI019 is an investigational drug under clinical development. Safety and efficacy have not been established.
CLBR001+SWI019 is a two-component infusion therapy of autologous switchable chimeric antigen receptor T (sCAR-T) cells (CLBR001) and cluster of differentiation antigen 19 (CD19)-targeted antibody Fab switch (SWI019) being investigated in relapsed or refractory (R/R) B-cell malignancies.1,2,3.
PROPOSED MECHANISM OF ACTION
The CLBR001+SWI019 sCAR-T system consists of two parts including an autologous cell product comprised of a humanized, third generation CAR-T cell (CLBR001) along with a humanized antibody Fab “switch” (SWI019) which is dosed following CLBR001 infusion. SWI019 is designed to act as a switch to dynamically turn “on” or “off” CLBR001 cells, allowing for the controllable activity of the adoptively transferred cells. CLBR001+SWI019 demonstrated target cell cytotoxicity in preclinical models.
DEVELOPMENT
CLBR001+SWI019 is being investigated in a Phase 1 trial for the treatment of R/R B-cell malignancies.
CLINICAL TRIALS
View select clinical trials with CLBR001+SWI019. To view a full list of clinical trials in which CLBR001+SWI019 is being investigated, please visit ClinicalTrials.gov.
References
- Young, T. CLBR001+SWI019: A novel switchable CAR-T cell platform enabling functionally reversible on/off control of CAR-T cell activity for B cell malignancies [abstract]. In: Proceedings of the American Association for Cancer Research Annual Meeting 2021; 2021 Apr 10-15 and May 17-21. Philadelphia (PA): AACR; Cancer Res 2021;81(13_Suppl):Abstract nr ND04.
- Nikolaenko L, et al. First in Human Study of an on/Off Switchable CAR-T Cell Platform Targeting CD19 for B Cell Malignancies (CLBR001+SWI019). Blood. 2021;138(1):2822.
- Laborda E, et al. IND-Enabling Studies of a Switchable Chimeric Antigen Receptor-T Cell (CLBR001+SWI019) to Support First in Human Clinical Study. Blood. 2021;138(1):1695.
OVERVIEW
Epcoritamab (ABBV-GEN3013)* is an approved drug being investigated for additional uses. Safety and efficacy have not been established for these additional uses.
Epcoritamab (ABBV-GEN3013) is an immunotherapy and an IgG1 bispecific antibody targeting CD3 and CD20.1,2
PROPOSED MECHANISM OF ACTION
Epcoritamab is a subcutaneously administered, bispecific CD3×CD20 antibody created via Fab-arm exchange using the unique DuoBody® technology platform that allows retaining of the native IgG1 antibody structure and a long plasma half-life.3
CD20 is a clinically well-established target that is expressed in a wide variety of B-cell malignancies. Epcoritamab binds to CD20 B-cells, and simultaneously to CD3 on T-cells, inducing activation and expansion of T-cells and T-cell mediated killing of CD20-positive malignant B cells.1,3
Upon the bispecific binding of CD3 on T cells and CD20 malignant B cells, an immunological synapse is formed between the bound T and B cell, resulting in perforin/granzyme B-induced apoptosis of the malignant B cells.1
®A registered trademark of Genmab A/S
DEVELOPMENT
Epcoritamab* FDA approval timeline9
- May 2023 (accelerated approval): For the treatment of adult patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL), not otherwise specified (NOS), including DLBCL arising from indolent lymphoma, and high grade B-cell lymphoma (HGBL) after 2 or more lines of systemic therapy.
This indication is approved under accelerated approval based on response rate and durability of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial(s).
AbbVie and Genmab continue to investigate Epcoritamab in ongoing Phase 3 and early phase clinical trials for the treatment of DLBLC, as well as other cancers, including follicular lymphoma (FL) and chronic lymphocytic leukemia (CLL).
EPCORITIMAB INDICATION AND SAFETY OVERVIEW FOR DLBCL (US PRESCRIBING INFORMATION)9
Indication:
Epcoritamab is a bispecific CD20-directed CD3 T-cell engager indicated for the treatment of adult patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL), not otherwise specified, including DLBCL arising from indolent lymphoma, and high-grade B-cell lymphoma after two or more lines of systemic therapy.
This indication is approved under accelerated approval based on response rate and durability of response.
Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial(s).
SAFETY INFORMATION
Boxed Warnings
- Cytokine release syndrome (CRS), including serious or life-threatening reactions, can occur in patients receiving Epcoritamab. Initiate treatment with the Epcoritamab step-up dosing schedule to reduce the incidence and severity of CRS. With hold Epcoritamab until CRS resolves or permanently discontinue based on severity.
- Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS), including life-threatening and fatal reactions, can occur with Epcoritamab. Monitor patients for neurological signs or symptoms of ICANS during treatment. Withhold Epcoritamab until ICANS resolves or permanently discontinue based on severity.
Warnings & Precautions
- Infections: Can cause serious or fatal infections. Monitor patients for signs or symptoms of infection, including opportunistic infections, and treat appropriately.
- Cytopenias: Monitor complete blood cell counts during treatment.
- Embryo-Fetal Toxicity: May cause fetal harm. Advise females of reproductive potential of the potential risk to the fetus and to use effective contraception.
Adverse Reactions
The most common (≥ 20%) adverse reactions are cytokine release syndrome, fatigue, musculoskeletal pain, injection site reactions, pyrexia, abdominal pain, nausea, and diarrhea. The most common Grade 3 to 4 laboratory abnormalities (≥ 10%) are decreased lymphocyte count, decreased neutrophil count, decreased white blood cell count, decreased hemoglobin, and decreased platelets.
Use in Specific Populations
Lactation: Advise not to breastfeed.
Review full prescribing information for additional information at: https://www.genmab-pi.com/prescribing-information/epkinly-pi.pdf
CLINICAL TRIALS
View select clinical trials with epcoritamab now. To view a full list of clinical trials in which epcoritamab is being investigated, please visit ClinicalTrials.gov.
- Hutchings M, et al. Epcoritamab (GEN3013; DuoBody-CD3×CD20) to induce complete response in patients with relapsed/refractory B-cell non-Hodgkin lymphoma: Complete dose escalation data and efficacy results from a phase I/II trial. Poster #8009. 56th Annual ASCO Meeting and Exposition; May 29 – June 2, 2020; Virtual Format.
- Engelberts PJ, et al. DuoBody-CD3xCD20 induces potent T-cell-mediated killing of malignant B cells in preclinical models and provides opportunities for subcutaneous dosing. EBioMedicine. 2020;52:102625.
- Hutchings M, et al. Dose escalation of subcutaneous epcoritamab in patients with relapsed or refractory B-cell non-Hodgkin lymphoma: an open-label, phase 1/2 study. Lancet. 2021;398(10306):1157-1169
- ClinicalTrials.gov. NCT03625037. https://clinicaltrials.gov/ct2/show/NCT03625037. Accessed October 2020.
- EudraCT Number 2020-000845-15. https://www.clinicaltrialsregister.eu/ctr-search/trial/2020-000845-15/DK/. Accessed November 2020.
- ClinicalTrials.gov. NCT04542824. https://clinicaltrials.gov/ct2/show/NCT04542824. Accessed October 2020.
- ClinicalTrials.gov. NCT04623541. https://clinicaltrials.gov/ct2/show/NCT04623541. Accessed November 2020.
- ClinicalTrials.gov. NCT04628494. https://clinicaltrials.gov/ct2/show/NCT04628494. Accessed November 2020.
- Epcoritamab [package insert]. North Chicago, IL; AbbVie, Inc. and Plainsboro, NJ: Genmab US, Inc.: May 2023.
OVERVIEW
Ibrutinib (PCI-32765, CRA-032765) is an approved drug being investigated for additional uses. Safety and efficacy have not been established for these additional uses.
Ibrutinib (PCI-32765, CRA-032765) is a selective, orally bioavailable small-molecule BTK inhibitor..1
PROPOSED MECHANISM OF ACTION
Ibrutinib (PCI-32765, CRA-032765) is small-molecule inhibitor of Bruton’s tyrosine kinase (BTK). Ibrutinib forms a covalent bond with a cysteine residue in the BTK active site, leading to inhibition of BTK enzymatic activity.1 BTK is a signaling molecule of the B-cell antigen receptor (BCR) and cytokine receptor pathways. BTK’s role in signaling through the B-cell surface receptors results in activation of pathways necessary for B-cell trafficking, chemotaxis, and adhesion. Nonclinical studies show that ibrutinib inhibits malignant B-cell proliferation and survival in vivo as well as cell migration and substrate adhesion in vitro.2
Ibrutinib also inhibits the activity of interleukin-2-inducible-T-cell kinase (ITK).3
DEVELOPMENT
Ibrutinib FDA approval timeline4
- Feb 2014 (accelerated approval): Monotherapy treatment of CLL or SLL patients who have received at least one prior therapy
- July 2014: Expanded indication to include patients with del(17p) CLL
- Jan 2015: Monotherapy for the treatment of adult patients with WM
- Mar 2016: Monotherapy for the treatment of previously untreated CLL
- May 2016: Ibrutinib in combination with BR for adult patients with previously-treated CLL/SLL
- Aug 2017: Monotherapy for the treatment of adult patients with cGVHD after failure of one or more lines of systemic therapy
- Aug 2018: Ibrutinib with rituximab for the treatment of previously untreated and relapsed/refractory WM
- Jan 2019: Ibrutinib plus obinutuzumab for the treatment of previously untreated CLL/SLL
- Apr 2020: Ibrutinib with rituximab for the treatment of previously untreated adults with CLL/SLL
- Aug 2022: Monotherapy for the treatment of pediatric patients 1 year and older with cGVHD after failure of one or more lines of systemic therapy
Imbruvica (ibrutinib) is jointly developed and commercialized by Pharmacyclics LLC, an AbbVie company and Janssen Biotech, Inc.
IBRUTINIB Indication and Safety Overview (US PRESCRIBING INFORMATION)5
Indication:
Ibrutinib is a kinase inhibitor indicated for the treatment of:
- Adult patients with chronic lymphocytic leukemia (CLL)/small lymphocytic lymphoma (SLL).
- Adult patients with chronic lymphocytic leukemia (CLL)/small lymphocytic lymphoma (SLL) with 17p deletion.
- Adult patients with Waldenström's macroglobulinemia (WM).
- Adult and pediatric patients age 1 year and older with chronic graft versus host disease (cGVHD) after failure of one or more lines of systemic therapy.
SAFETY INFORMATION
Warning and Precautions
- Hemorrhage: Fatal bleeding events have occurred in patients who received ibrutinib. Major hemorrhage (≥ Grade 3, serious, or any central nervous system events; e.g., intracranial hemorrhage [including subdural hematoma], gastrointestinal bleeding, hematuria, and post procedural hemorrhage) occurred in 4.2% of patients, with fatalities occurring in 0.4% of 2,838 patients who received ibrutinib in 27 clinical trials. Bleeding events of any grade including bruising and petechiae occurred in 39%, and excluding bruising and petechiae occurred in 23% of patients who received ibrutinib, respectively.
The mechanism for the bleeding events is not well understood.
Use of either anticoagulant or antiplatelet agents concomitantly with ibrutinib increases the risk of major hemorrhage. Across clinical trials, 3.1% of 2,838 patients who received ibrutinib without antiplatelet or anticoagulant therapy experienced major hemorrhage. The addition of antiplatelet therapy with or without anticoagulant therapy increased this percentage to 4.4%, and the addition of anticoagulant therapy with or without antiplatelet therapy increased this percentage to 6.1%. Consider the risks and benefits of anticoagulant or antiplatelet therapy when co-administered with ibrutinib. Monitor for signs and symptoms of bleeding.
Consider the benefit-risk of withholding ibrutinib for at least 3 to 7 days pre- and post-surgery depending upon the type of surgery and the risk of bleeding. - Infections: Fatal and non-fatal infections (including bacterial, viral, or fungal) have occurred with ibrutinib therapy. Grade 3 or greater infections occurred in 21% of 1,476 patients with B-cell malignancies who received ibrutinib in clinical trials. Cases of progressive multifocal leukoencephalopathy (PML) and Pneumocystis jirovecii pneumonia (PJP) have occurred in patients treated with ibrutinib. Consider prophylaxis according to standard of care in patients who are at increased risk for opportunistic infections. Monitor and evaluate patients for fever and infections and treat appropriately.
- Cardiac Arrhythmias, Cardiac Failure, and Sudden Death: Fatal and serious cardiac arrhythmias and cardiac failure have occurred with ibrutinib. Deaths due to cardiac causes or sudden deaths occurred in 1% of 4,896 patients who received ibrutinib in clinical trials, including in patients who received ibrutinib in unapproved monotherapy or combination regimens. These adverse reactions occurred in patients with and without preexisting hypertension or cardiac comorbidities. Patients with cardiac comorbidities may be at greater risk of these events.
Grade 3 or greater ventricular tachyarrhythmias were reported in 0.2%, Grade 3 or greater atrial fibrillation and atrial flutter were reported in 3.7%, and Grade 3 or greater cardiac failure was reported in 1.3% of 4,896 patients who received ibrutinib in clinical trials, including in patients who received ibrutinib in unapproved monotherapy or combination regimens. These events have occurred particularly in patients with cardiac risk factors including hypertension and diabetes mellitus, a previous history of cardiac arrhythmias, and in patients with acute infections.
Evaluate cardiac history and function at baseline, and monitor patients for cardiac arrhythmias and cardiac function. Obtain further evaluation (e.g., ECG, echocardiogram) as indicated for patients who develop symptoms of arrhythmia (e.g., palpitations, lightheadedness, syncope, chest pain), new onset dyspnea, or other cardiovascular concerns. Manage cardiac arrhythmias and cardiac failure appropriately, follow dose modification guidelines, and consider the risks and benefits of continued ibrutinib treatment. - Hypertension: Hypertension occurred in 19% of 1,476 patients with B-cell malignancies who received ibrutinib in clinical trials. Grade 3 or greater hypertension occurred in 8% of patients. Based on data from a subset of these patients, (N=1,124), the median time to onset was 5.9 months (range, 0 to 24 months). In a long-term safety analysis over 5 years of 1,284 patients with B-cell malignancies treated for a median of 36 months (range, 0 to 98 months), the cumulative rate of hypertension increased over time. The prevalence for Grade 3 or greater hypertension was 4% (year 0-1), 7% (year 1-2), 9% (year 2-3), 9% (year 3-4), and 9% (year 4-5); the overall incidence for the 5-year period was 11%. Monitor blood pressure in patients treated with ibrutinib, initiate or adjust anti-hypertensive medication throughout treatment with ibrutinib as appropriate, and follow dosage modification guidelines for Grade 3 or higher hypertension.
- Cytopenias: In 645 patients with B-cell malignancies who received ibrutinib as a single agent, grade 3 or 4 neutropenia occurred in 23% of patients, grade 3 or 4 thrombocytopenia in 8% and grade 3 or 4 anemia in 2.8%, based on laboratory measurements. Monitor complete blood counts monthly.
- Second Primary Malignancies: Other malignancies (10%), including non-skin carcinomas (3.9%), occurred among the 1,476 patients with B-cell malignancies who received ibrutinib in clinical trials. The most frequent second primary malignancy was non-melanoma skin cancer (6%).
- Tumor Lysis Syndrome: Tumor lysis syndrome has been infrequently reported with ibrutinib. Assess the baseline risk (e.g., high tumor burden) and take appropriate precautions. Monitor patients closely and treat as appropriate.
- Embryo-Fetal Toxicity: Based on findings in animals, ibrutinib can cause fetal harm when administered to a pregnant woman. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with ibrutinib and for 1 month after the last dose. Advise males with female partners of reproductive potential to use effective contraception during the same time period.
Adverse Reactions
- B-cell malignancies: The most common adverse reactions (≥30%) in adult patients with B-cell malignancies were thrombocytopenia (55%)*, diarrhea (44%), fatigue (39%), musculoskeletal pain (39%), neutropenia (39%)*, rash (36%), anemia (35%)*, bruising (32%), and nausea (30%).
The most common Grade ≥ 3 adverse reactions (≥5%) in adult patients with B-cell malignancies were neutropenia (21%)*, thrombocytopenia (14%)*, pneumonia (8%), and hypertension (8%).
Approximately 9% (CLL/SLL), and 14% (WM) of adult patients had a dose reduction due to adverse reactions. Approximately 4-10% (CLL/SLL) and 5% (WM) of adult patients discontinued due to adverse reactions. - cGVHD: The most common adverse reactions (≥20%) in adult or pediatric patients with cGVHD were fatigue (57%), anemia (49%)*, bruising (40%), diarrhea (36%), thrombocytopenia (33%)*, musculoskeletal pain (30%), pyrexia (30%), muscle spasms (29%), stomatitis (29%), hemorrhage (26%), nausea (26%), abdominal pain (23%), pneumonia (23%), and headache (21%).
The most common Grade 3 or higher adverse reactions (≥5%) reported in adult or pediatric patients with cGVHD were pneumonia (14%), anemia (13%)*, fatigue (12%), pyrexia (11%), diarrhea (10%), neutropenia (10%)*, sepsis (10%), osteonecrosis (9%), stomatitis (9%), hypokalemia (7%), headache (5%), and musculoskeletal pain (5%).
Discontinuation of ibrutinib treatment due to an adverse reaction occurred in 24% of adult patients and 23% of pediatric patients. Adverse reactions leading to dose reduction occurred in 26% of adult patients and 19% of pediatric patients.
*Treatment-emergent decreases (all grades) were based on laboratory measurements.
Drug Interactions
- CYP3A Inhibitors: Co-administration of ibrutinib with strong or moderate CYP3A inhibitors may increase ibrutinib plasma concentrations. Increased ibrutinib concentrations may increase the risk of drug-related toxicity. Dose modifications of ibrutinib are recommended when used concomitantly with posaconazole, voriconazole, and moderate CYP3A inhibitors. Avoid concomitant use of other strong CYP3A inhibitors. Interrupt ibrutinib if strong inhibitors are used short-term (e.g., for ≤ 7 days). Avoid grapefruit and Seville oranges during ibrutinib treatment, as these contain strong or moderate inhibitors of CYP3A. See dose modification guidelines in USPI sections 2.3 and 7.1.
- CYP3A Inducers: Avoid coadministration with strong CYP3A inducers.
Specific Populations
- Pediatric Use: The safety and effectiveness of ibrutinib have not been established for the treatment of cGVHD after failure of one or more lines of therapy in pediatric patients less than 1 year of age. The safety and effectiveness of ibrutinib in pediatric patients have not been established in CLL/SLL, CLL/SLL with 17p deletion, WM, or in patients with mature B-cell non-Hodgkin lymphoma.
- In the randomized population from a study that included 35 patients (26 pediatric patients age 5 to less than 17 years) with previously treated mature B-cell non-Hodgkin lymphoma, major hemorrhage and discontinuation of chemoimmunotherapy due to adverse reactions occurred more frequently in the ibrutinib plus chemoimmunotherapy arm compared to the chemoimmunotherapy alone arm.
- Hepatic Impairment:
- Adult Patients with B-cell Malignancies: Hepatic Impairment (based on Child-Pugh criteria): Avoid use of ibrutinib in patients with severe hepatic impairment. In patients with mild or moderate impairment, reduce recommended ibrutinib dose and monitor more frequently for adverse reactions of ibrutinib.
- Patients with cGVHD: Avoid use of ibrutinib in patients with total bilirubin level > 3x upper limit of normal (ULN) (unless of non-hepatic origin or due to Gilbert’s syndrome). Reduce recommended dose when administering ibrutinib to patients with total bilirubin level > 1.5 to 3x ULN (unless of non-hepatic origin or due to Gilbert’s syndrome).
Review full US prescribing information for additional information at www.rxabbvie.com.
CLINICAL TRIALS
View select clinical trials with ibrutinib now. To view a full list of clinical trials in which venetoclax is being investigated, please visit ClinicalTrials.gov.
- Honigberg LA, Smith AM, Sirisawad M, et al. The Bruton tyrosine kinase inhibitor PCI-32765 blocks B-cell activation and is efficacious in models of autoimmune disease and B-cell malignancy. Proc Natl Acad Sci USA. 2010 Jul 20 ; 107(29) :13075-80.
- Herman SEM, Gordon AL, Hertlein E, et al. Bruton tyrosine kinase represents a promising therapeutic target for treatment of chronic lymphocytic leukemia and is effectively targeted by PCI-32765. Blood. 2011 Jun 9 ;117(23) :6287-96.
- Dubovsky JA, Flynn R, Du J, et al. Ibrutinib treatment ameliorates murine chronic graft-versus-host disease. J Clin Invest. 2014 Nov;124(11):4867-76.
- Drugs@FDA: FDA Approved Drug Products – IMBRUVICA. Accessed at https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&ApplNo=208573
- Ibrutinib [package insert]. South San Francisco, CA: Pharmacyclics LLC, and Horsham, PA: Janssen Biotech, Inc.: 5/2023.