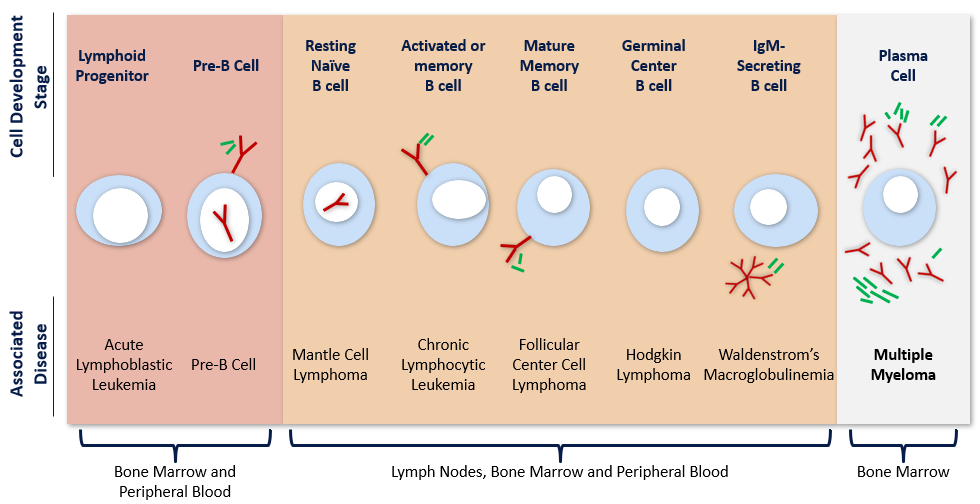
MULTIPLE MYELOMA (MM)
Exploring epigenetic modulation and inducing apoptosis as potential
therapeutic approaches for patients with multiple myeloma.1-3
- Bui MH, Lin X, Albert DH, et al. Preclinical characterization of BET family bromodomain inhibitor ABBV-075 suggests combination therapeutic strategies. Cancer Res. 2017;77(11):2976-2989.
- Kumar S, Kaufman JL, Gasparetto C, et al. Efficacy of venetoclax as targeted therapy for relapsed/refractory t(11;14) multiple myeloma. Blood. 2017 Oct 10. [Epub ahead of print]
- Moreau P, Chanan-Khan A, Roberts AW, et al. Promising efficacy and acceptable safety of venetoclax plus bortezomib and dexamethasone in relapsed/refractory MM. Blood. 2017 Aug 28. [Epub ahead of print]
- Cowan AJ, et al. Global Burden of Multiple Myeloma. JAMA Oncol. 2018;4(9):1221-1227.
- Howlader N, Noone AM, Krapcho M, Miller D, Brest A, Yu M, Ruhl J, Tatalovich Z, Mariotto A, Lewis DR, Chen HS, Feuer EJ, Cronin KA (eds). SEER Cancer Statistics Review, 1975-2017, National Cancer Institute. Bethesda, MD, https://seer.cancer.gov/csr/1975_2017/, based on November 2019 SEER data submission, posted to the SEER web site, April 2020.
- Radovic VV. J Med Biochem. 2010;29:1-8.
- Rajkumar SV, Kumar S. Multiple myeloma: diagnosis and treatment. Mayo Clin Proc. 2016;91(1):101-119.
- Zhang B, Gojo I, Fenton RG. Myeloid cell factor-1 is a critical survival factor for multiple myeloma. Blood. 2002;99(6):1885-1893.
- Kyrtsonis MC, Bartzis V, Papanikolaou X, et al. Genetic and molecular mechanisms in multiple myeloma: a route to better understand disease pathogenesis and heterogeneity. Appl Clin Genet. 2010;3:41-51.
- Punnoose EA, et al. Expression Profile of BCL-2, BCL-XL, and MCL-1 Predicts Pharmacological Response to the BCL-2 Selective Antagonist Venetoclax in Multiple Myeloma Models. Mol Cancer Ther. 2016;15(5):1132-1144.
- Trudel S, et al. The Bcl-2 Family Protein Inhibitor, ABT-737, Has Substantial Antimyeloma Activity and Shows Synergistic Effect with Dexamethasone and Melphalan. Clin Cancer Res. 2007;13(2):621-629.
- Touzeau C, et al. The Bcl-2 specific BH3 mimetic ABT-199: a promising targeted therapy for t(11;14) multiple myeloma. Leukemia. 2014;28:210-2012.
- Rajkumar SV, et al. International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol. 2014;15(12):e538-48.
- Lakshman, A., et al. Natural history of t(11;14) multiple myeloma. Leukemia 2017, 32(1), 131-138.
- Palumbo A, Avet-Loiseau H, Oliva S, et al. Revised international staging system for multiple myeloma: a report from International Myeloma Working Group. J Clin Oncol. 2015;33(26):2863-2869.
- Durer C, et al. Treatment of relapsed multiple myeloma: Evidence-based recommendations. Blood Rev. 2020;39:100616.
- Chim CS, et al. Management of relapsed and refractory multiple myeloma: novel agents, antibodies, immunotherapies and beyond. Leukemia. 2018;32(2):252-262.
- Maes A, Menu E, Veirman K, et al. The therapeutic potential of cell cycle targeting in multiple myeloma. Oncotarget. 2017;8(52):90501-90520.
- Paszekova, et al. High-Risk Multiple Myeloma: Different Definitions, Different Outcomes? Clinical Lymphoma, Myeloma and Leukemia, 2014;14(1):24-30.
- Bal S, et al. New regimens and directions in the management of newly diagnosed multiple myeloma. Am J Hematol. 2021;96(3):367-378.
- Larocca A, Mina R, Gay F, Bringhen S, Boccadoro M. Emerging drugs and combinations to treat multiple myeloma. Oncotarget. 2017;8(36):60656-60672.
- NCI. Cancer Stat Facts: Myeloma. https://seer.cancer.gov/statfacts/html/mulmy.html. Accessed August 2023.
- Global Burden of Disease Cancer Collaboration; Sung H; Ferlay J, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA: Cancer J. Clin. 2021;71(3):209-249.
- Rajkumar SV. Multiple myeloma: 2020 update on diagnosis, risk-stratification and management. Am J Hematol. 2020;95(5):548-567.
- Rajkumar SV, Kumar S. Multiple myeloma current treatment algorithms. Blood Cancer J. 2020 Sep 28;10(9):94.
- Yong K, Delforge M, Driessen C, et al. Multiple myeloma: patient outcomes in real-world practice. Br J Haematol. 2016;175(2):252-264.
- D’Agostino M, et al. Second Revision of the International Staging System (R2-ISS) for Overall Survival in Multiple Myeloma: A European Myeloma Network (EMN) Report Within the HARMONY Project. J Clin Oncol. 2022 Oct 10;40(29):3406-3418.