CD3 x CD20 BISPECIFICS
OVERVIEW
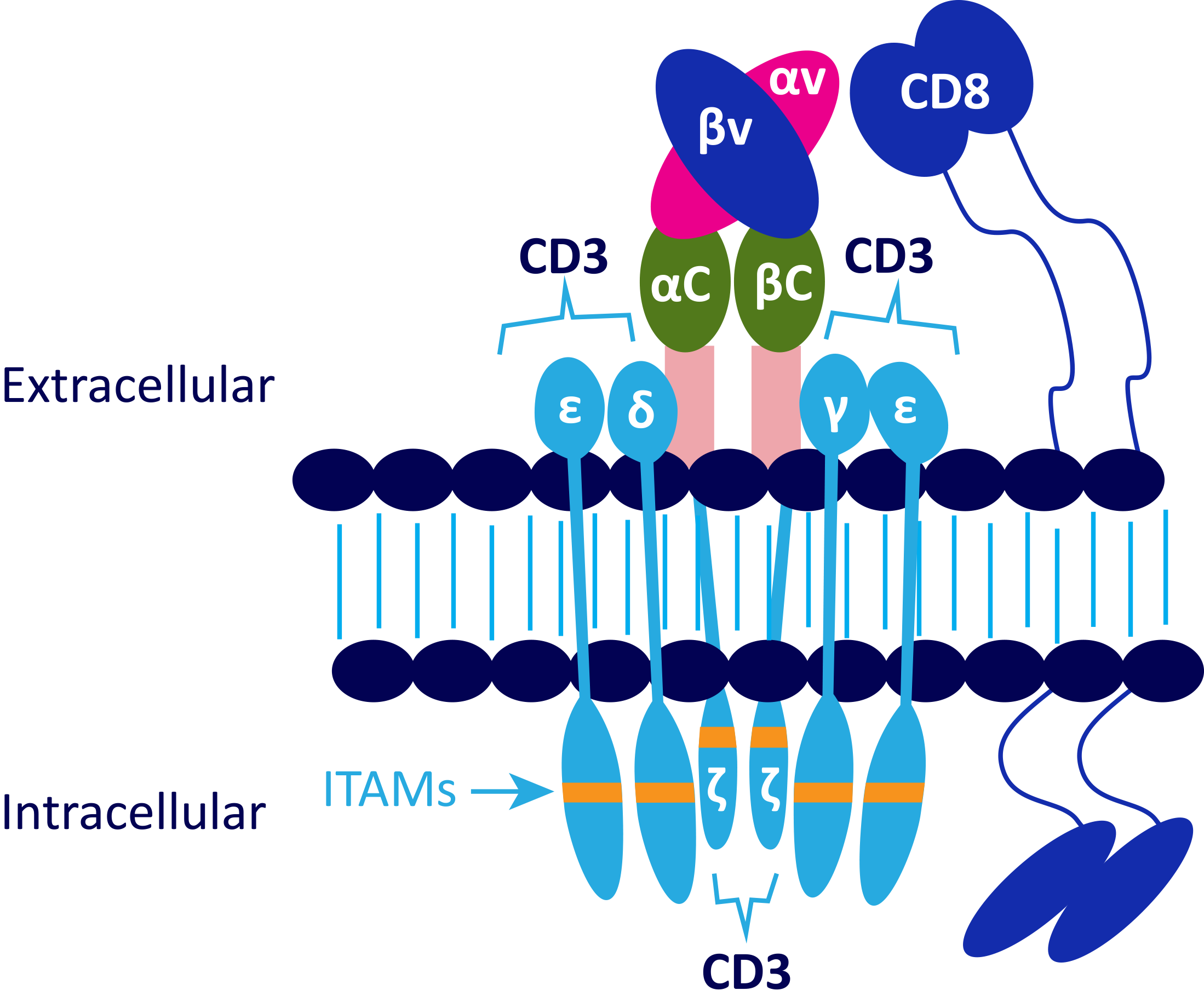
T cell receptors (TCRs) are protein complexes formed by six different polypeptides. The CD3 (cluster of differentiation 3) complex is part of the T cell receptor (TCR) complex and is composed of two CD3ε, one CD3γ, one CD3δ, and two CD3ζ chains.1,2 CD3 complex ensures signal transduction and plays an important role in T cell activation.1-3
CD20 (cluster of differentiation 20) is a membrane-embedded surfaced molecule which plays a role in the development and differentiation of B cells into plasma cells.4 CD20 on B cells is a clinically well-validated target that is expressed in a wide variety of B cell malignancies with limited normal tissue expression beyond B -cells.5,6
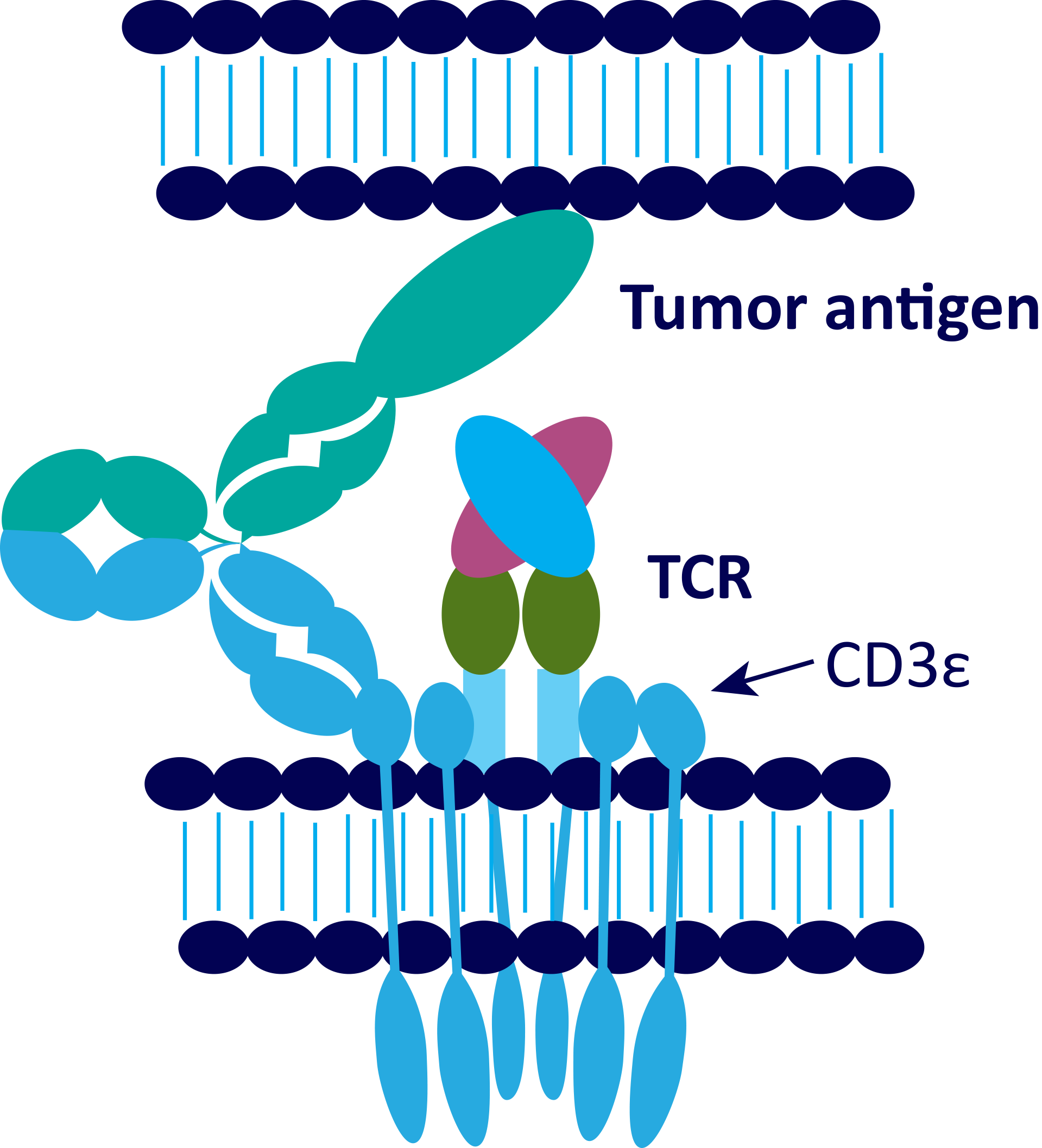
A CD3 x CD20 bispecific antibody binds to CD20 on malignant cells, while simultaneously binding to CD3 on T cells, inducing activation and cytotoxic activity of T cells enabling lysis of CD20-expressing malignant cells.1,4,7
Diagram of the T-cell receptor (TCR) complex.
- CD3 bispecific antibodies (bsAb) induce the activation and cytotoxic activity of effector T cells enabling lysis of target-expressing malignant cells.1,3,7
- CD3 bsAb activate T cells by binding CD3 on T cells and a tumor-associated antigen (TAA), such as CD20, on the cancer cells. Consequently, available T cells can bind to target-expressing cells irrespective of the peptide/major histocompatibility complex (MHC) specificity of their T cell receptor (TCR).1,3
- Activation of the TCR-CD3 complexes leads to the formation of a cytolytic synapse between the T cell and the tumor cell which results in the release of perforin and granzymes and subsequent tumor cell death by apoptosis.1
T cell redirection with bispecific antibodies in which one binding arm recognizes a tumor antigen and the other binding arm recognizes CD3 on T cells.
- CD3 bsAbs in clinical trials either lack an Fc region or contain an engineered Fc domain to minimize interaction with Fc receptors.1,3
- The presence of an Fc domain increases the in vivo half-life through binding to the neonatal Fc receptor.3
- A key CD3 bsAb development consideration is a need for a format that either significantly limits or eliminates cyokine release syndrome (CRS), since CRS appears to be dose-limiting in most cases for this class of molecules.1
IMPLICATIONS IN CANCER
Immunotherapy of cancer with CD3 bsAb is a fast developing field, and multiple TAAs are under evaluation for both hematological and solid tumor malignancies with many different CD3 bsAbs in clincial development.3,7 The idea of using the cytotoxic capacity of T cells through CD3 bsAb to kill tumor cells dates back to 1980’s.3,8
CD20 receptor is a well-established target expressed by multiple B cell malignancies. Resistance is rarely caused by loss of CD20 expression as evidenced by the presence of CD20 on B cells in the vast majority of patients previously treated with CD20-containing regimens.6
Bispecific antibodies that bind to both CD3 and CD20 can recruit T cells from the body’s own immune system to act as cytotoxic effectors against tumor cells.6
Related Research
- Strohl WR, Naso M. Bispecific T-Cell Redirection versus Chimeric Antigen Receptor (CAR)-T Cells as Approaches to Kill Cancer Cells. Antibodies (Basel). 2019 Jul 3;8(3):41.
- Alcover A, et al. Cell Biology of T Cell Receptor Expression and Regulation. Annu. Rev. Immunol. 2018. 36:85–107.
- Benonisson H, et al. CD3-Bispecific Antibody Therapy Turns Solid Tumors into Inflammatory Sites but Does Not Install Protective Memory. Mol Cancer Ther. 2019;18(2):312-322.
- Ratcliffe MJ, et al. Encyclopedia of Immunobiology. 2016.
- Hutchings M, et al. Dose escalation of subcutaneous epcoritamab in patients with relapsed or refractory B-cell non-Hodgkin lymphoma: an open-label, phase 1/2 study. Lancet. 2021;398(10306):1157-1169.
- Engelberts PJ, et al. DuoBody-CD3xCD20 induces potent T-cell-mediated killing of malignant B cells in preclinical models and provides opportunities for subcutaneous dosing. EBioMedicine. 2020 Feb;52:102625.
- Labrijn AF, Janmaat ML, Reichert JM, Parren PWHI. Bispecific antibodies: a mechanistic review of the pipeline. Nat Rev Drug Discov. 2019 Aug;18(8):585-608.
- Staerz UD, Kanagawa O, Bevan MJ. Hybrid antibodies can target sites for attack by T cells. Nature. 1985 Apr 18-24;314(6012):628-31.