MYELOFIBROSIS
Exploring dysfunctional pathways, mechanisms, and biomarkers in
myelofibrosis to discover new insights into the progression of the disease.
- Mehta J, et al. Epidemiology of myeloproliferative neoplasms in the United States. Leukemia & Lymphoma. 2014;55(3):595-600.
- Shallis RM, et al. Epidemiology of the classical myeloproliferative neoplasms: The four corners of an expansive and complex map. Blood Reviews. 2020 Jul;42:100706. DOI:10.1016/j.blre.2020.100706.
- Gangat N, et al. DIPSS Plus: A Refined Dynamic International Prognostic Scoring System for Primary Myelofibrosis That Incorporates Prognostic Information From Karyotype, Platelet Count, and Transfusion Status. Journal of Clinical Oncology. 2011;29(4):392-397.
- Howlader N, et al.SEER Cancer Statistics Review1975-2017; https://seer.cancer.gov/csr/1975_2017/browse_csr.php?sectionSEL=30&pageSEL=sect_30_table.01.. Accessed November 2020.
- https://seer.cancer.gov/explorer/application.html?site=409&data_type=1&graph_type=2&compareBy=sex&chk_sex_1=1&race=1&age_range=1&hdn_stage=101&hdn_rate_type=1&advopt_precision=1&advopt_display=2
- https://www.medpagetoday.com/resource-centers/polycythemia-vera/incidence-myeloproliferative-neoplasms-population-based-us-data/1155
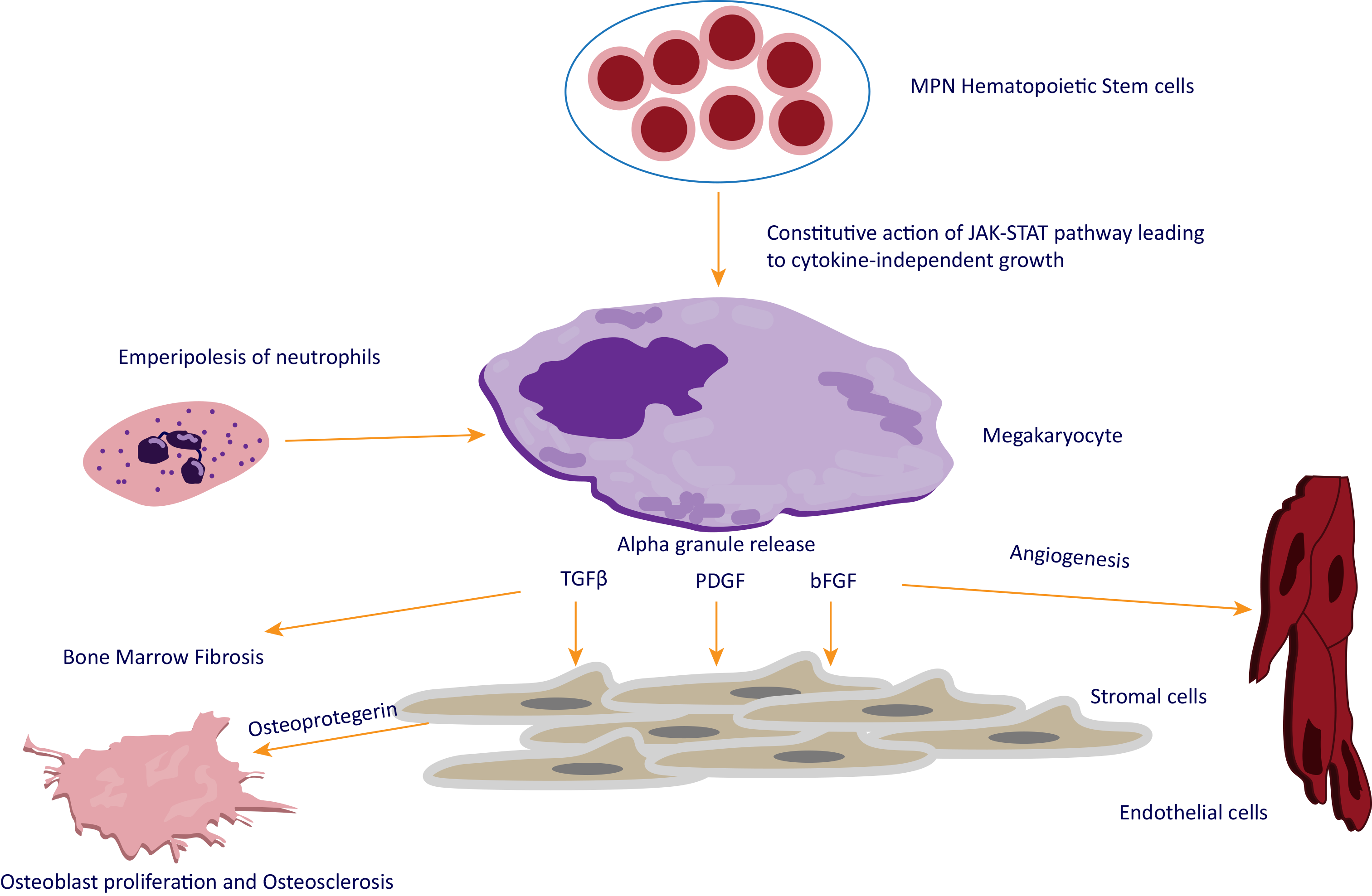
- Agarwal A, et al. Bone marrow fibrosis in primary myelofibrosis: pathogenic mechanisms and the role of TGF-β. Stem Cell Investig 2016;3:5. (image)
- Barbui T, et al. The 2016 WHO classification and diagnostic criteria for myeloproliferative neoplasms: document summary and in-depth discussion. Blood Cancer J. 2018;8(2):15.
- Malara A, et al. Megakaryocyte Contribution to Bone Marrow Fibrosis: many Arrows in the Quiver. Mediterr J Hematol Infect Dis. 2018;10(1):e2018068.
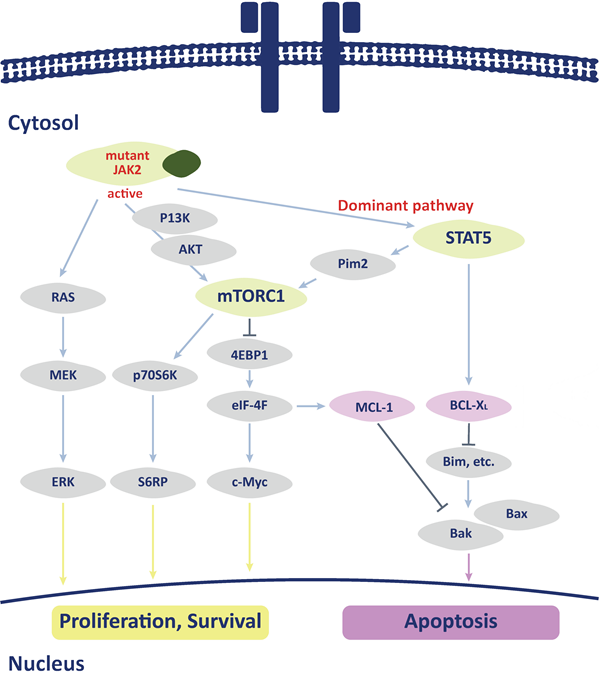
- Winter P, et al. RAS signaling promotes resistance to JAK inhibitors by suppressing BAD-mediated apoptosis. Science Signaling 2014. 7(357):ra122.
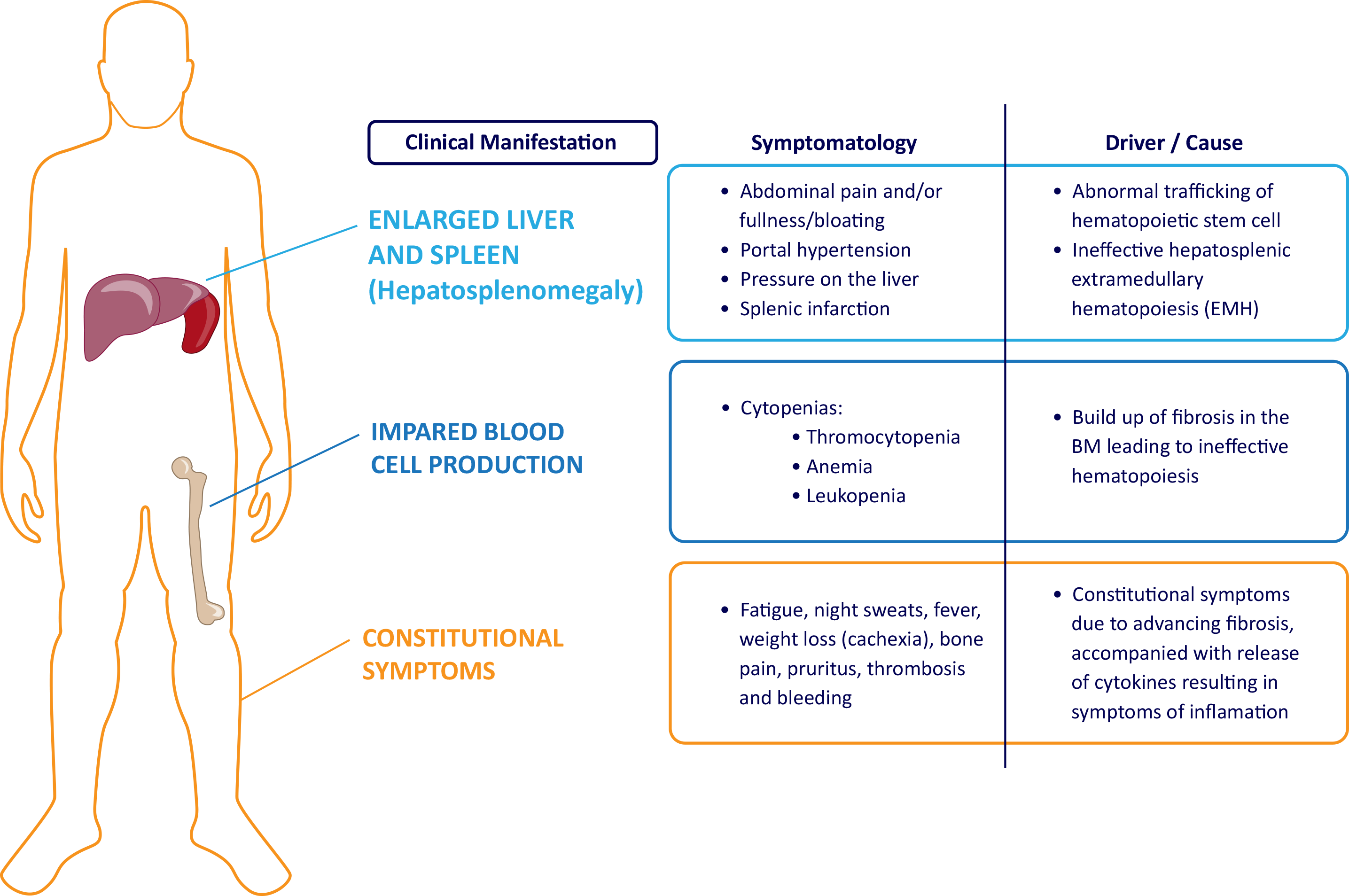
- Tefferi A, et al. Primary myelofibrosis: 2021 update on diagnosis, risk-stratification and management. Am J Hematol. 2021;96:145–162.
- The Leukemia & Lymphoma Society. Myelofibrosis Facts. https://www.lls.org/sites/default/files/file_assets/FS14_Myelofibrosis_Fact%20Sheet_Final9.12.pdf. Updated April 2012. Accessed November 2020.
- Alvarez-Larran A, et al. Postpolycythaemic myelofibrosis: frequency and risk factors for this complication in 116 patients. Br J Haematol 2009;146(5):504-509.
- Tefferi A. Essential thrombocythemia, polycythemia vera, and myelofibrosis: current management and the prospect of targeted therapy. Am J Hematol. 2008;83:491-497.
- Liu C, Hao S. (2017). 'Chapter 8 – Primary Myelofibrosis', in Chang C-C, Ohgami RS.(ed.) Precision Molecular Pathology of Myeloid Neoplasms. Springer, pp. 155-179.
- Tefferi A, et al. Leukemia risk models in primary myelofibrosis: an International Working Group study. Leukemia (2012) 26, 1439–1441.
- Cervantes F, et al. New prognostic scoring system for primary myelofibrosis based on a study of the International Working Group for Myelofibrosis Research and Treatment. Blood . 2009 Mar 26;113(13):2895-901.
- Passamonti F, Cervantes F, Vannucchi AM, et al. A dynamic prognostic model to predict survival in primary myelofibrosis: a study by the IWG-MRT (International Working Group for Myeloproliferative Neoplasms Research and Treatment). Blood 2010;115:1703-1708.
- Waibel M, et al. Combined Targeting of JAK2 and Bcl-2/Bcl-xL to Cure Mutant JAK2-Driven Malignancies and Overcome Acquired Resistance to JAK2 Inhibitors. Cell Rep. 2013;5(4):1047-1059.
- Zhang M, et al. Selective targeting of JAK/STAT signaling is potentiated by Bcl-xL blockade in IL-2–dependent adult T-cell leukemia. Proc Natl Acad Sci U S A. 2015;112(40):12480-12485.
- De Freitas RM, et al. Myeloproliferative neoplasms and the JAK/STAT signaling pathway: an overview. Rev Bras Hematol Hemoter. 2015;37(5):348–353.
- Zeuner A, et al. Activity of the BH3 mimetic ABT-737 on polycythemia vera erythroid precursor cells. Blood. 2009;113(7):1522-5.