CD39
OVERVIEW
Ecto-enzyme CD39 (ectonucleoside triphosphate diphosphohydrolase 1) is encoded by ETPD1 and is expressed by various immune and non-immune cells (e.g. fibroblasts and endothelial cells).1
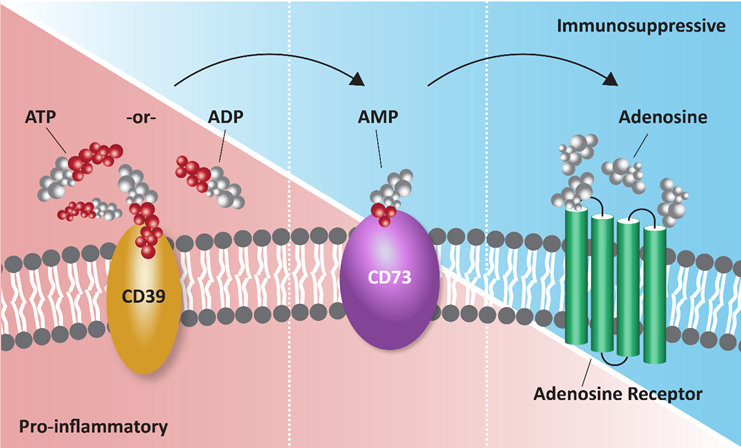
- The function of CD39 is to hydrolyze extracellular adenosine triphosphate (eATP) to adenosine monophosphate (AMP) by removing up to two phosphate groups. AMP can then be converted to extracellular adenosine by CD73, also an ecto-enzyme.1
Extracellular ATP acts as a pro-inflammatory molecule and is actively released from activated or stressed cells during inflammation, hypoxia or apoptosis and passively from necrotic cells via rupture.2 eATP binds to purinergic (P2) receptors widely expressed on immune and non-immune cells which initiates signaling cascades resulting in calcium mobilization, actin polymerization, chemotaxis, release of mediators, cell maturation, cytotoxicity, and death to induce an immune response.3
Extraceullar adenosine has the opposite effect, producing anti-inflammatory effects by binding P1 receptors (A1, A2A, A2B, and A3) which are also widely expressed, especially on immune cells.2
The ATP-adenosine pathway plays a key role in establishing an immunosuppressive tumor microenvironment (TME) by driving the conversion of proinflammatory, extracellular ATP to immunosuppressive adenosine.1
IMPLICATIONS IN CANCER
CD39 is constitutively expressed on immune cell populations, such as B cells, natural killer cells, dendritic cells, monocytes, macrophages, neutrophils, activated T cells, and regulatory T cells (Tregs), as well as endothelial cells.5 Further, some tumors express CD39 and intratumoral immune cells display elevated CD39 expression.1 CD39 is the rate-limiting enzymatic step in the production of immunosuppressive adenosine, and can be viewed as an immunological switch that shifts ATP-driven pro-inflammatory immune cell activity toward an anti-inflammatory, immunosuppressed state mediated by adenosine.5
- Extracellular adenosine acting at A2A receptors may have a major role in down-modulating the immune response as cancerous tissues contain elevated levels of adenosine and adenosine break-down products.4
- Targeting CD39 could encourage immune-mediated anti-tumor response via two mechanisms:5
- Increasing the availability of immunostimulatory extracellular ATP released by damaged and/or dying cells.
- Reducing the generation and accumulation of suppressive adenosine within the TME.
Related Research
- Moesta AK, et al. Targeting CD39 in cancer. Nature Reviews Immunology. 2020 Dec;20(12):739-55.
- Faas MM, et al. Extracellular ATP and adenosine: the Yin and Yang in immune responses?. Molecular Aspects of Medicine. 2017 Jun 1;55:9-19.
- Jacob F, et al. Purinergic signaling in inflammatory cells: P2 receptor expression, functional effects, and modulation of inflammatory responses. Purinergic signalling. 2013 Sep 1;9(3):285-306.
- Pellegatti P, et al. Increased level of extracellular ATP at tumor sites: in vivo imaging with plasma membrane luciferase. PLoS One 2008;3:e2599.
- Spatola BN, et al. Fully human anti-CD39 antibody potently inhibits ATPase activity in cancer cells via uncompetitive allosteric mechanism. InMAbs 2020 Jan 1 (Vol. 12, No. 1, p. 1838036). Taylor & Francis.