PD-1
OVERVIEW
Programmed death protein 1 (PD-1) is a type 1 cell surface receptor expressed by activated CD4+ and CD8+ T cells, natural killer (NK) T cells, and antigen-presenting cells (APCs).1,2
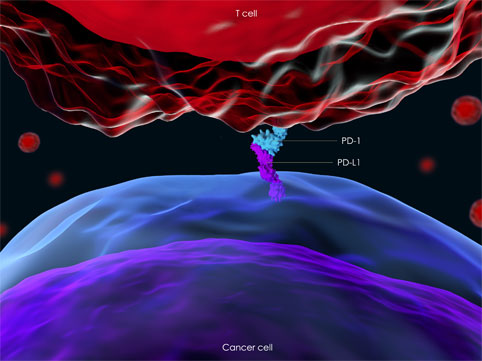
- The PD-1 receptor has two ligands, programmed death ligand-1 (PD-L1) and programmed death ligand-2 (PD-L2), which are mainly expressed on APCs. Receptor/ligand interactions maintain immune homeostasis by reducing T-cell priming and inhibiting effector T-cell proliferation and function while increasing immunosuppressive regulatory T cell (Treg) function.1,3,4
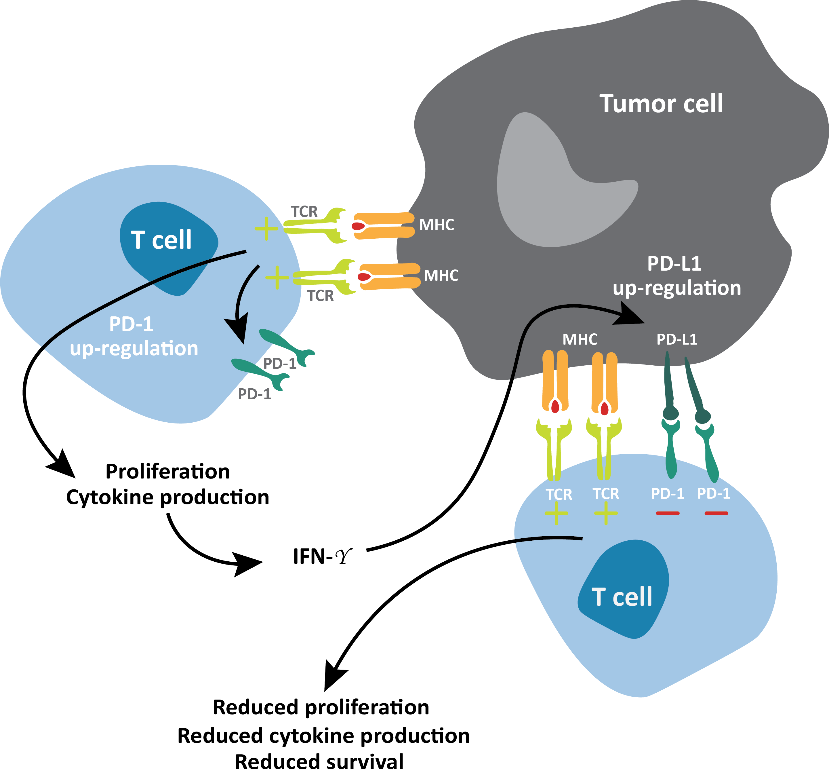
- T-cell activation by APCs via stimulation of the T-cell receptor (TCR) by major histocompatibility complexes (MHCs), costimulated by CD28 interactions with CD80 or CD86, upregulates the expression of PD-1 on those T cells.5,6,20
- Prolonged TCR stimulation during an ongoing immune response can cause upregulated PD-1 expression.20
- Tumor cells can express PD-L1 (and PD-L2, not shown) as a consequence of inflammatory cytokines and/or oncogenic signaling pathways. PD-1:PD-L1 binding inhibits TCR-mediated positive signaling, leading to reduced proliferation, reduced cytokine secretion, and reduced survival, thus down-regulating the immune response.20
IMPLICATIONS IN CANCER
- Appropriation of the PD-L1/PD-1 pathway can be one mechanism by which the tumor evades immunosurveillance.7
- Induction of PD-L1 expression on tumor cells, tumor-infiltrating APCs, and/or immune cells within the tumor microenvironment leads to suppression of immune responses and poor anti-tumor responses, permitting cancer progression and metastasis.8,9
- Immunohistochemistry (IHC) has also detected PD-L1 expression in infiltrating lymphocytes in a variety of tumors.10
- Effective cancer immunotherapy depends on the status of the immune response in the tumor microenvironment and patients with tumors that contain a high density of tumor-infiltrating lymphocytes (TILs) are most likely to respond to immunotherapy, although this is not sufficient for complete cancer eradication.
- Evidence of T-cell exhaustion has been observed in T cells infiltrating many solid tumors.11
- PD-L1 protein is commonly overexpressed in solid tumors.10
- In general, improved response to PD1/PD-L1 inhibitors tends to correlate with patient tumors having:8,18,23,29
- Higher PD-L1 protein expression
- Higher mutational load or tumor mutational burden (TMB)
- Higher mismatch repair deficiency (MMR) and microsatellite instability (MSI)
- Earlier line of therapy
- Many kinds of genomic aberrations such as copy number amplification of the genes PD-L1 and JAK2 can result in high expression of PD-L1 protein and poor prognosis; however, actual response to PD-1/PD-L1 antagonistic inhibition does not necessarily follow this biomarker pattern.18,23
MELANOMA
- Patients with melanoma have shown around 40-50% ORR to PD-1/PD-L1 antagonism irrespective of PD-L1 protein expression.23
- For melanoma, the promises that PD-L1 expression in tumor samples could serve as a predictive marker have been disappointing.24
- Instead, melanoma response to anti-PD-1 therapy correlates more with mutational load, which tends to be high in melanoma.24,25
NSCLC
- Anti-PD-1/PD-L1 therapies for NSCLC tend to use PD-L1 protein expression to select patients as response rates are higher in PD-L1-expressing tumors relative to unselected populations.23,26
- In general, PD-1/PD-L1 inhibiting therapies show more benefit in smokers versus non-smokers, however evidence suggests that the higher ORR is likely due to the higher tumor mutational burden associated with the smoking population.26
- Randomized phase III trials have indicated that patients carrying EGFR or ALK mutations exhibit less efficacy of anti-PD-1/PD-L1 treatment than those with wild-type, whereas KRAS mutations are likely to be predictors of favorable outcomes.18,30
HNSCC
- A statistically significant increase in ORR was observed in patients with HNSCC treated with a PD-1 inhibitor in PD-L1-positive (22%) versus PD-L1-negative (4%).23
- In HNSCC, smoking seems to contribute to a more immunosuppressive tumor microenvironment (TME) and negatively impact anti-PD-1/PD-L1 efficacy.31
- Increased TMB and neoantigen load have been shown to correlate with response to anti-PD-1/PD-L1 therapies in HPV- HNSCC, whereas most of the studies conducted to date have refuted their predictive value in HPV+ patients.31
RESISTANCE
- Up to 60% of patients across different tumor types display primary resistance to anti-PD-1/PD-L1 agents.31
- Several mechanisms have been suggested such as poor tumor immunogenicity, limited intratumoral immune cell infiltration, coexpression of multiple inhibitory receptors, and induction of immunosuppressive pathways within the TME.31
- To overcome the resistance, many ongoing clinical trials are evaluating combination strategies with other immunotherapies, targeted agents, chemotherapy and radiotherapy.31
Related Research
- Ohaegbulam KC, Assal A, Lazar-Molnar E, Yao Y, Zang X. Human cancer immunotherapy with antibodies to the PD-1 and PD-L1 pathway. Trends Mol Med. 2015;21(1):24-33.
- Gao J, Bernatchez C, Sharma P, Radvanyi LG, Hwu P. Advances in the development of cancer immunotherapies. Trends Immunol. 2013;34(2):90-98.
- Freeman GJ, Long AJ, Iwai Y, et al. Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J Exp Med. 2000;192(7):1027-1034.
- Latchman Y, Wood CR, Chernova T, et al. PD-L2 is a second ligand for PD-1 and inhibits T cell activation. Nat Immunol. 2001;2(3):261-268.
- Hui E, Cheung J, Zhu J, et al. T cell costimulatory receptor CD28 is a primary target for PD-1-mediated inhibition. Science. 2017;355(6332):1428-1433.
- O'Donnell JS, Smyth MJ, Teng MWL. PD1 functions by inhibiting CD28-mediated co-stimulation. Clin Transl Immunology. 2017;6(5):e138.
- Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646-674.
- O'Donnell JS, Long GV, Scolyer RA, Teng MW, Smyth MJ. Resistance to PD1/PDL1 checkpoint inhibition. Cancer Treat Rev. 2017;52:71-81.
- D'Incecco A, Andreozzi M, Ludovini V, et al. PD-1 and PD-L1 expression in molecularly selected non-small-cell lung cancer patients. Br J Cancer. 2015;112(1):95-102.
- Kythreotou A, Siddique A, Mauri FA, Bower M, Pinato DJ. PD-L1. J Clin Pathol. 2018;71(3):189-194.
- Ahmadzadeh M, Johnson LA, Heemskerk B, et al. Tumor antigen-specific CD8 T cells infiltrating the tumor express high levels of PD-1 and are functionally impaired. Blood. 2009;114(8):1537-1544.
- Scheel AH, Ansén S, Schultheis AM, et al. PD-L1 expression in non-small cell lung cancer: Correlations with genetic alterations. Oncoimmunology. 2016;5(5):e1131379.
- Sheng J, Fang W, Yu J, et al. Expression of programmed death ligand-1 on tumor cells varies pre and post chemotherapy in non-small cell lung cancer. Sci Rep. 2016;6:20090.
- Azuma K, Ota K, Kawahara A, et al. Association of PD-L1 overexpression with activating EGFR mutations in surgically resected nonsmall-cell lung cancer. Ann Oncol. 2014;25(10):1935-1940.
- Kaunitz GJ, Cottrell TR, Lilo M, et al. Melanoma subtypes demonstrate distinct PD-L1 expression profiles. Lab Invest. 2017;97(9):1063-1071.
- Hino R, Kabashima K, Kato Y, et al. Tumor cell expression of programmed cell death-1 ligand 1 is a prognostic factor for malignant melanoma. Cancer. 2010;116(7):1757-1766.
- Hamanishi J, Mandai M, Iwasaki M, et al. Programmed cell death 1 ligand 1 and tumor-infiltrating CD8+ T lymphocytes are prognostic factors of human ovarian cancer. Proc Natl Acad Sci U.S.A. 2007;104(9):3360-3365.
- Yi M, Jiao D, Xu H, et al. Biomarkers for predicting efficacy of PD-1/PD-L1 inhibitors. Mol Cancer. 2018;17(1):129.
- Sharma P, Callahan MK, Bono P, et al. Nivolumab monotherapy in recurrent metastatic urothelial carcinoma (CheckMate 032): a m ulticenter, open-label, two-stage, multi-arm, phase 1/2 trial. Lancet Oncol. 2016;17(11):1590-1598.
- Buchbinder EI, Desai A. CTLA-4 and PD-1 Pathways: Similarities, Differences, and Implications of Their Inhibition. Am J Clin Oncol. 2016;39(1):98-106.
- Garces AHI, et al. Building on the anti-PD1/PD-L1 backbone: combination immunotherapy for cancer. Expert Opinion on Investigational Drugs. 2019;28(8):695-708.
- Yan Y, et al. Combining immune checkpoint inhibitors with conventional cancer therapy. Front. Immunol. 2018; 9:1739.
- Chen Q, et al. Drug response to PD-1/PD-L1 blockade: based on biomarkers. OncoTargets and Therapy. 2018;11:4673-4683.
- Heinzerling L, et al. Predicting the response to anti-PD1 therapy in metastatic melanoma. Transl Cancer Res. 2016;5(S3):S576-S579.
- Marconcini R, et al. Current status and perspectives in immunotherapy for metastatic melanoma. Oncotarget. 2018;9(15):12452-12470.
- Califano R, et al. Patient selection for anti-PD-1/PD-L1 therapy in advanced non-small-cell lung cancer: implications for clinical practice. Future Oncol. 2018;14(23):2415-2431.
- Nowicki TS, et al. Mechanisms of Resistance to PD-1 and PD-L1 blockade. Cancer J. 2018;24(1):47-53.
- Simon S and N Labarriere. PD-1 expression on tumor-specific T cells: Friend or foe for immunotherapy? Oncoimmunology. 2018;7(1):e1364828 (7 pages).
- Gong J, et al. Development of PD-1 and PD-L1 inhibitors as a form of cancer immunotherapy: a comprehensive review of registration trial and future considerations. Journal for ImmunoTherapy of Cancer. 2018;6:8.
- Miura Y and N Sunaga. Role of Immunotherapy for Oncogene-Driven Non-Small Cell Lung Cancer. Cancers. 2018;10:245.
- Oliva M, et al. Immune biomarkers of response to immunecheckpoint inhibitors in head and neck squamous cell carcinoma. Annals of Oncology. 2019;30:57-67.