IMMUNO-ONCOLOGY
OVERVIEW
The human body has many natural immune mechanisms for cancer cell recognition and elimination. Normal function of these effector mechanisms helps prevent tumor growth and metastasis.1-3 Unfortunately, counter mechanisms can also be established that allow malignant cells to evade this destruction by the immune system.3-6
The discovery and development of immuno-oncology therapy represents a milestone in the treatment of cancer. Unlike traditional therapies that focus on attacking the tumor cells themselves, immuno-oncology is a revolutionary treatment approach which focuses on remobilizing the patient's own immune system to recognize and eliminate cancer cells.5-8
Normal Immune Response
There are two main components of the immune response: innate and adaptive immunity.1,2,6These are complementary networks of self-defense against non-self, foreign threats to the human body.1,2,6
Innate immune response1,2,6
- An antigen-independent response that is immediate and has no immunologic memory
- Natural killer (NK) cells9-11, dendritic cells (DCs), innate lymphoid cells (ILCs), and phagocytes (macrophages and neutrophils) are the primary innate immune cell types.
Adaptive immune response1,2,6
- An antigen-dependent and antigen-specific response with the capacity for immunologic memory
- T cells and B cells are the primary adaptive immune cell types.
- Macrophages, B cells, and DCs activate T cells via antigen cross presentation (AP).1
- Unlike T cells, B cells can recognize free antigen directly, without the need for antigen presenting cells (APCs).2
- B cells are converted to plasma cells to kill organisms via secreted antibodies, which is referred to as antibody-dependent cell-mediated cytotoxicity.1
Role of the Immune System in Cancer
Tumor development can potentially be controlled by cytotoxic innate and adaptive immune cells; however, as the tumor develops from neoplastic tissue to clinically detectable tumors, cancer cells evolve different mechanisms that mimic peripheral immune tolerance in order to avoid tumoricidal attack.27 The innate and adaptive immune response has the ability to detect and eliminate these early tumor cells that are no longer recognized as normal self (abnormal) via tumor antigens.1-3
Potential malfunctions in immunity leading to tumor progression include:6
- Failure of tumor antigen recognition
- DCs and T cells may treat antigens as self rather than foreign
- T cells may not properly localize to tumors
- T cells may not effectively infiltrate the tumor microenvironment
- Factors in the tumor microenvironment might suppress effector cells
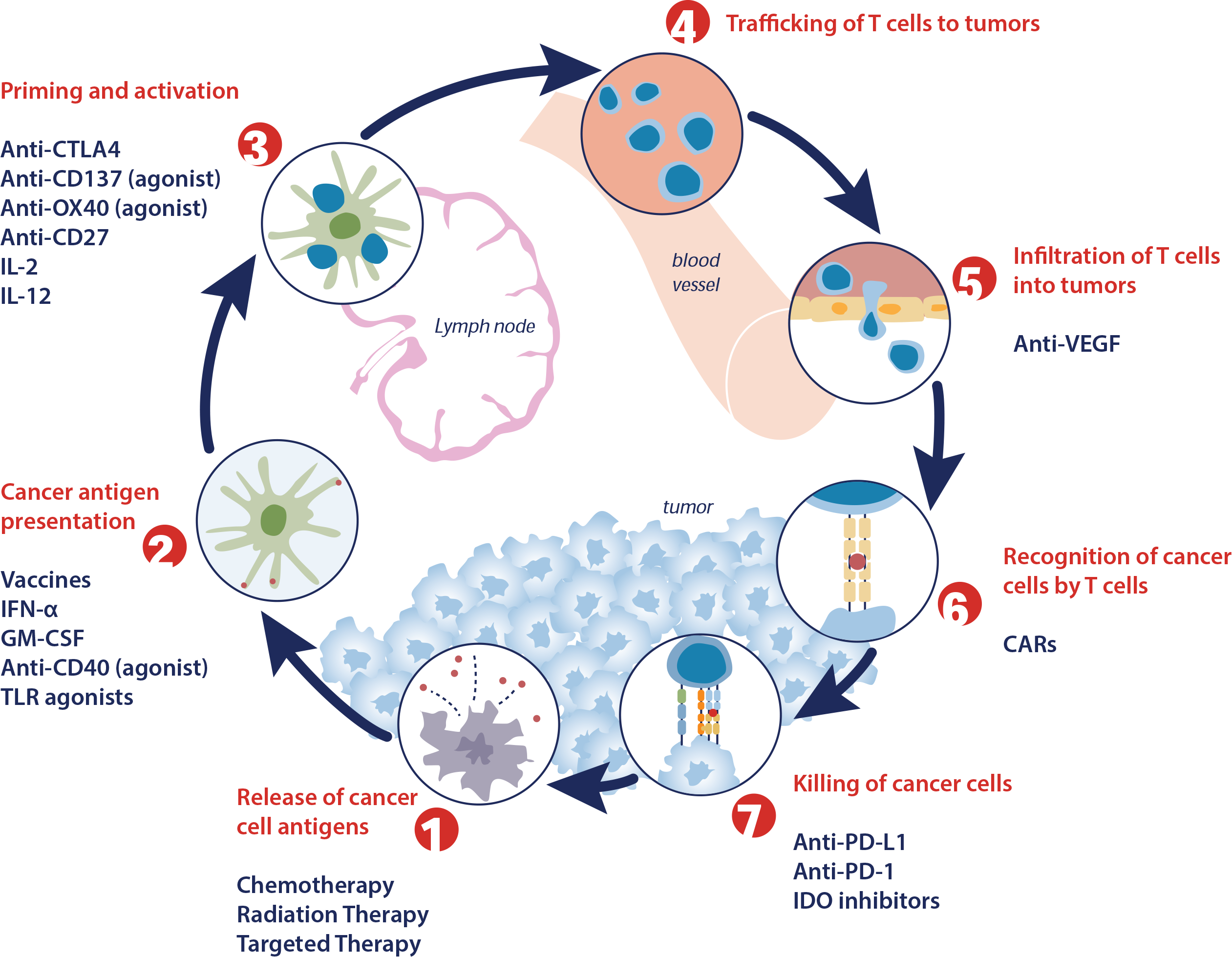
Steps in the Cancer Immunity Cycle / Opportunities for Anti-Cancer Therapeutics
- Release of Cancer Antigens
- Damage- or Pathogen- associated molecular pattern (DAMP/PAMP) molecules released from microbes or infected, stressed or dying cells (including cancer cells) activate innate immune cells which are capable of rapidly responding to the threat.11-13
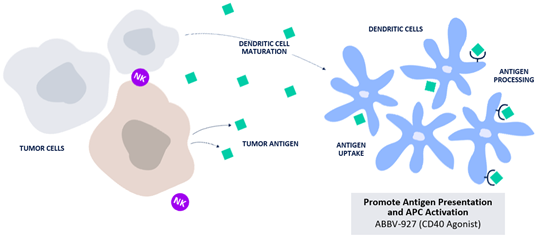
- These same danger signals activate and mature DCs, the first step in the adaptive immune response.12,13
- Cancer Antigen Presentation
- Antigen presentation to T cells by activated and mature DCs or APCs.2,5
- Mature DCs capture and process proteins from the threat (antigens for the adaptive immune system to recognize) and migrate to draining lymph nodes.14
- Major histocompatibility complex (MHC) proteins on the surface of mature DCs present the captured antigens to the T-cell receptor (TCR) on T cells.2,6 This, along with the costimulation, is required for T-cell activation.
- Antigen presentation to T cells by activated and mature DCs or APCs.2,5
- T-cells secrete other cytokines to further control the immune response and activate B-cells that multiply and mature into antibody-producing plasma cells.2
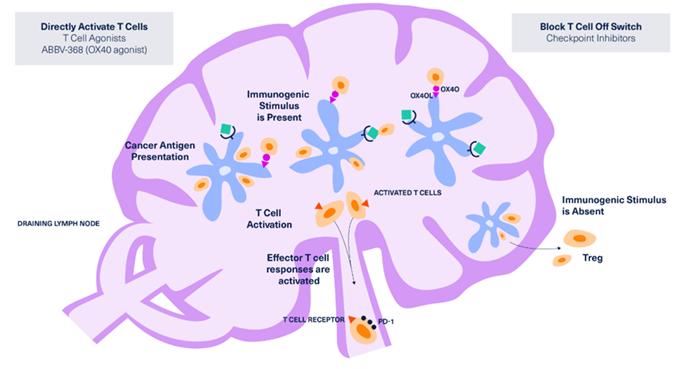
3. Priming and Activation of T Cells
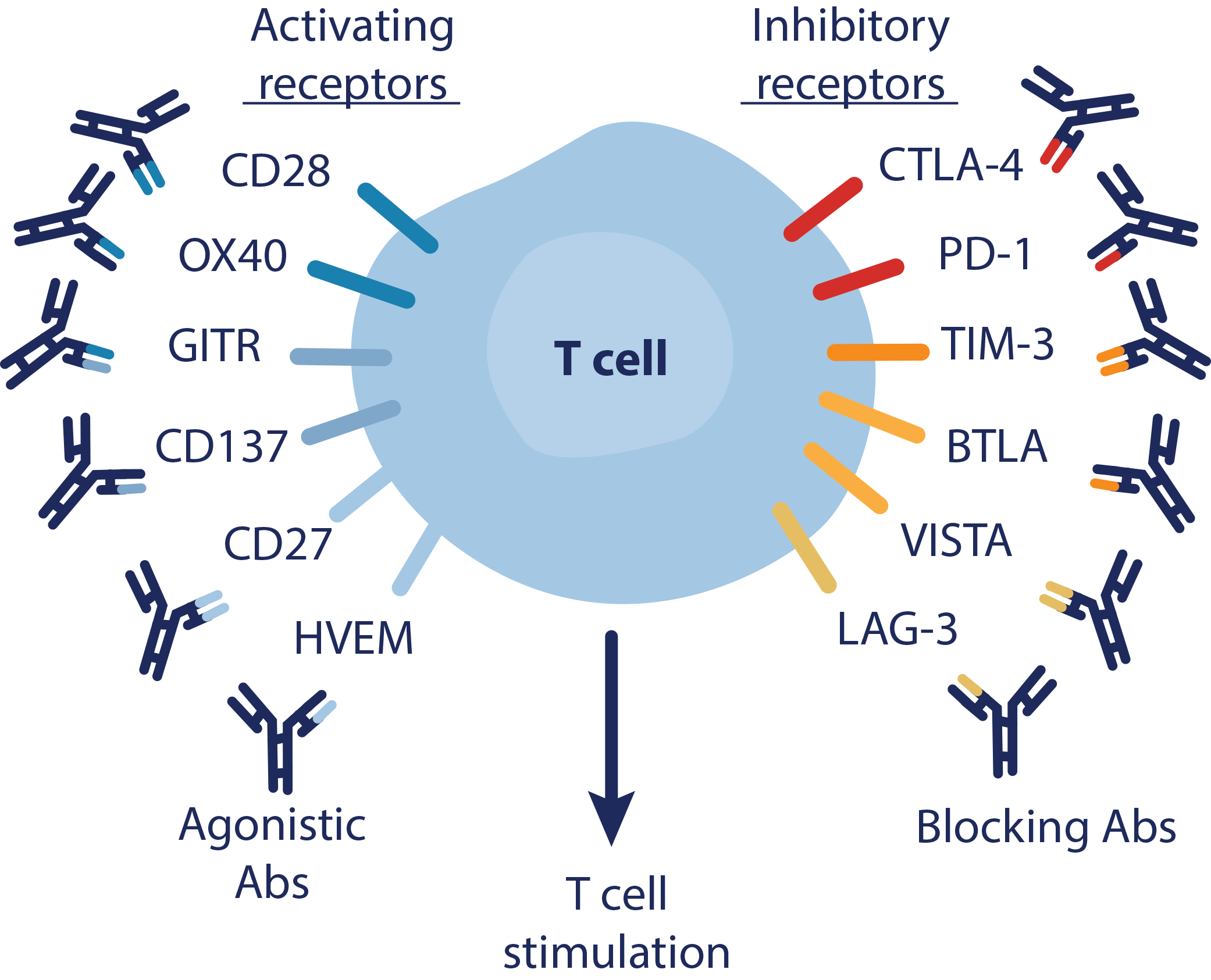
- The balance between CD8+/CD4+ T cells (effectors) and T-cell regulators (Tregs) within a tumor is important in determining the response to anti-PD1/PD-L1 agents and other cancer immunotherapy.6
- Stimulators of T-cell priming and activation include cytokines and surface receptors such as OX40, CD28, GITR, IL-2, IL-12, CD137, and CD27.6,17
- Inhibitors (checkpoints) include CTLA4/B7.1, PD-1/PD-L1/B7.1, prostaglandins, TIM3, LAG3, TIGIT.6,10,18,19
4. Trafficking of T Cells to Tumors
- The trafficking of activated T cells and other immune cells to the tumor is a highly regulated and dynamic process, involving a series of distinct processes, which include rolling, adhesion, extravasation, and chemotaxis.5,20
- Overcoming barriers that restrict T-cell infiltration to the tumor site is critical.
- Stimulators include CX3CL1, CXCL9, CXCL10, and CCL5.6,17
5. Infiltration of T Cells into Tumors
- T-cells, as well as macrophages and monocytes, must infiltrate the tumor microenvironment.6
- Stimulators include LFA1:ICAM1, selectins.5,6
- Immunosuppressive defense mechanisms produced by the tumor include upregulation of PD-L1/L2 and release of VEGF.6,17
6. Recognition of Cancer Cells by T cells
- The T-cell receptor (TCR) specifically recognizes and binds to the corresponding tumor antigen.5,6
- Response may be reduced if the cancer cell:14
- Has lost expression of the tumor antigen
- Has downregulated their expression of MHC class I molecules
- Expresses surface molecules (ie, PD-L1) that engage receptors on activated T cells (ie, PD-1), leading to T-cell exhaustion
Immunosuppression and Immunotherapy Resistance
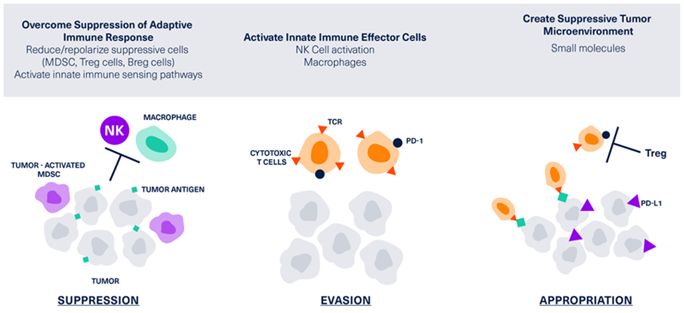
For tumors to grow, mechanisms must be in place to evade the immune response that might recognize and eliminate the tumor cells.3,22
Suppression, evasion, and appropriation of natural immune effector mechanisms for tumor detection and elimination contribute to tumor growth and metastasis.3,4
- Suppression of these mechanisms prevents the natural antitumor immune response.3,22
- Evasion of these mechanisms occurs via changes in the tumor cells that prevent recognition by immune cells, such as:21
- Loss of cell surface tumor antigens
- Loss of sensitivity to complement, T-cell, or natural killer (NK)-cell induced lysis
- Appropriation of normal immune functions occurs within the tumor microenvironment3
Many tumor-cell–intrinsic and –extrinsic factors contribute to immune evasion and immuno-oncology therapy resistance.4
Intrinsic Mechanisms of Resistance
- Intrinsic mechanisms are those that involve the tumor cell itself.
- Mechanisms of primary resistance include alteration of signaling pathways (ie, JAK, MAPK, PIK3, WNT), lack or loss of tumor-specific antigens, alteration in antigen-presenting machinery, constitutive PD-L1 expression, and loss of human leukocyte antigen (HLA) expression.4
- Mechanisms of acquired resistance include loss of target antigen or HLA expression at recurrence, altered IFN signaling, loss of T-cell functionality, and loss of sensitivity to complement-induced, T cell-induced, or natural killer (NK) cell-induced lysis.4
Extrinsic Mechanisms of Resistance
- Extrinsic mechanisms involve components of the tumor microenvironment other than the tumor cell.
- Extrinsic mechanisms of primary and adaptive resistance to immunotherapy include absence of T cells, inhibitory immune checkpoints, and immunosuppressive cells.4,8,22,23
Therapeutic Potential
The introduction of immune checkpoint inhibitors in oncology has increased the potential for durable responses and shifted therapeutic attention to extending the tail of the survival curve. However, not all patients respond to currently available immunotherapy agents (ie, PD-1/PD-L1 and CTLA-4 antibodies).5,15,18
The potential to address a range of additional immune mechanisms of resistance and to activate an immune response at additional points along the immune cycle makes continued research in immuno-oncology vital to the evolution of cancer therapy. Novel immuno-oncology targets and pathways include OX40 and CD40.14,17,23,24
Due to the multiple mechanisms of immune-suppression, combination immuno-oncology regimens may also help address patient needs.
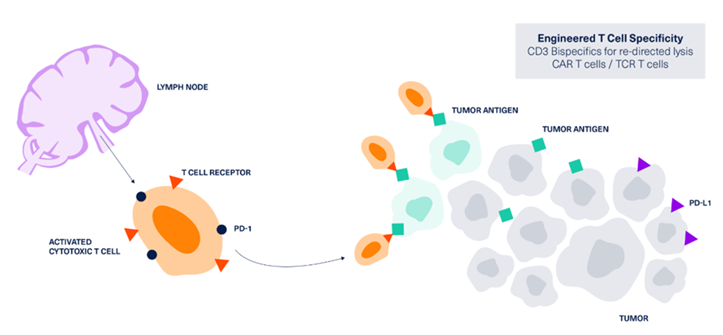
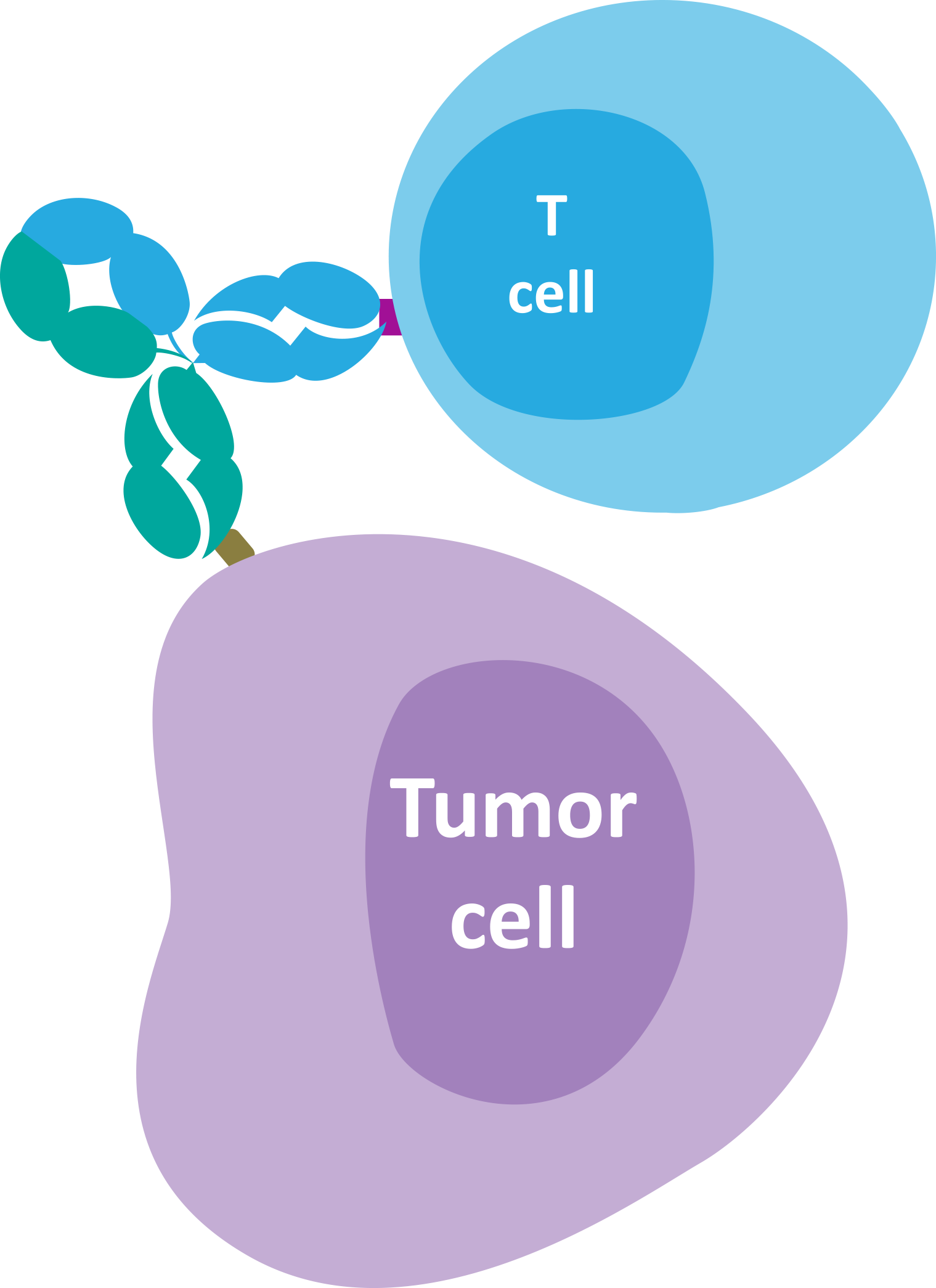
One additional emerging anti-tumor strategy utilizing the immune system is the concept of T-cell redirecting bispecific antibodies (TRBAs). These types of bispecific antibodies can simultaneously bind to a target on T cells and a target on malignant cells, inducing activation and cytotoxic activity of T cells enabling tumor cell death.7,25,26
The ongoing research into immuno-oncology therapies is opening up exciting new avenues, exemplified by the impressive therapeutic response observed with checkpoint inhibitors targeting PD-1/L1 and CTLA-4.27
- Zhang Q, et al. Resolution of Cancer-Promoting Inflammation: A New Approach for Anticancer Therapy. Front. Immunol. 2017;8:71.
- Warrington R, et al. An introduction to immunology and immunopathology. Allergy Asthma Clin Immunol. 2011;7 Suppl 1:S1.
- Finn OJ. Immuno-oncology: understanding the function and dysfunction of the immune system in cancer. Ann Oncol. 2012;23(suppl 8):viii6-viii9.
- Sharma P, Hu-Lieskovan S, Wargo JA, Ribas A. Primary, adaptive, and acquired resistance to cancer immunotherapy. Cell. 2017;168(4):707-723.
- Kim JM, Chen DS. Immune escape to PD-L1/PD-1 blockade: seven steps to success (or failure). Ann Oncol. 2016;27(8):1492-1504.
- Chen DS, Mellman I. Oncology meets immunology: the cancer-immunity cycle. Immunity. 2013;39(1):1-10. (image)
- Lowe KL, et al. Novel TCR-based biologics: mobilising T cells to warm 'cold' tumours. Cancer Treatment Reviews. 2019;77:35–43.
- Pan C, et al. Next-generation immuno-oncology agents: current momentum shifts in cancer immunotherapy. Journal of Hematology & Oncology. 2020;13:29.
- Gismondi A, et al. Effector functions of natural killer cell subsets in the control of hematological malignancies. Front. Immunol. 2015; 6:567.
- Marcus A, et al. Recognition of tumors by the innate immune system and natural killer cells. Adv Immunol. 2014;122:91-128.
- Liu Y and G Zeng. Cancer and innate immune system interactions: translational potentials for cancer immunotherapy. J Immunother. 2012;35(4):299-308.
- Hernandez C, et al. Damage-associated molecular patterns in cancer: a double-edged sword. Oncogene. 2016;35(46):5931-5941.
- Ferguson TA, et al. Armed response: how dying cells influence T-cell functions. Immunol Rev. 2011;241(1):77-88.
- Mellman I, et al. Cancer immunotherapy comes of age. Nature. 2011;480(7378):480-489.
- Chen DS, et al. Molecular pathways: next-generation immunotherapy–inhibiting programmed death-ligand 1 and programmed death-1. Clin Cancer Res. 2012;18(24)6580-6587. (image)
- Wieczorek M, et al. Major histocompatibility complex (MHC) class I and MHC class II proteins: conformational plasticity in antigen presentation. Front Immunol. 2017;8:292.
- Franciszkiewicz K, et al. CD103 or LFA-1 engagement at the immune synapse between cytotoxic T cells and tumor cells promotes maturation and regulates T-cell effector functions. Cancer Res. 2013;73(2):617-628.
- Riella LV, et al. Role of the PD-1 pathway in the immune response. Am J Transplant. 2012;12(10):2575-2587.
- So T, et al. Tumor necrosis factor/tumor necrosis factor receptor family members that positively regulate immunity. Int J Hematol. 2006;83(1):1-11.
- Slaney CY, et al. Trafficking of T cells into tumors. Cancer Res. 2014;74(24):7168-7174.
- Fan G, Wang Z, Hao M, Li J. Bispecific antibodies and their applications. J Hermatol Oncol. 2015;8:130.
- Wang L, et al. Connecting blood and intratumoral Treg cell activity in predicting future relapse in breast cancer.
- Gabrilovich DI. Myeloid-derived suppressor cells. Cancer Immunol Res. 2017;5(1):3-8.
- Bell RB, et al. OX40 signaling in head and neck squamous cell carcinoma: Overcoming immunosuppression in the tumor microenvironment. Oral Oncology. 2016;52:1–10. (image)
- Strohl WR and M Naso. Bispecific T-Cell Redirection versus Chimeric Antigen Receptor (CAR)-T Cells as Approaches to Kill Cancer Cells. Antibodies. 2019;8: 41. doi:10.3390/antib8030041.
- Hutchings M, et al. Epcoritamab (GEN3013; DuoBody-CD3×CD20) to induce complete response in patients with relapsed/refractory B-cell non-Hodgkin lymphoma: Complete dose escalation data and efficacy results from a phase I/II trial. Poster #8009. 56th Annual ASCO Meeting and Exposition; May 29 – June 2, 2020; Virtual Format.
- Gonzalez H, et al. Roles of the immune system in cancer: from tumor initiation to metastatic progression. Genes Dev. 2018;32(19-20):1267-1284.