c-MET Protein
OVERVIEW
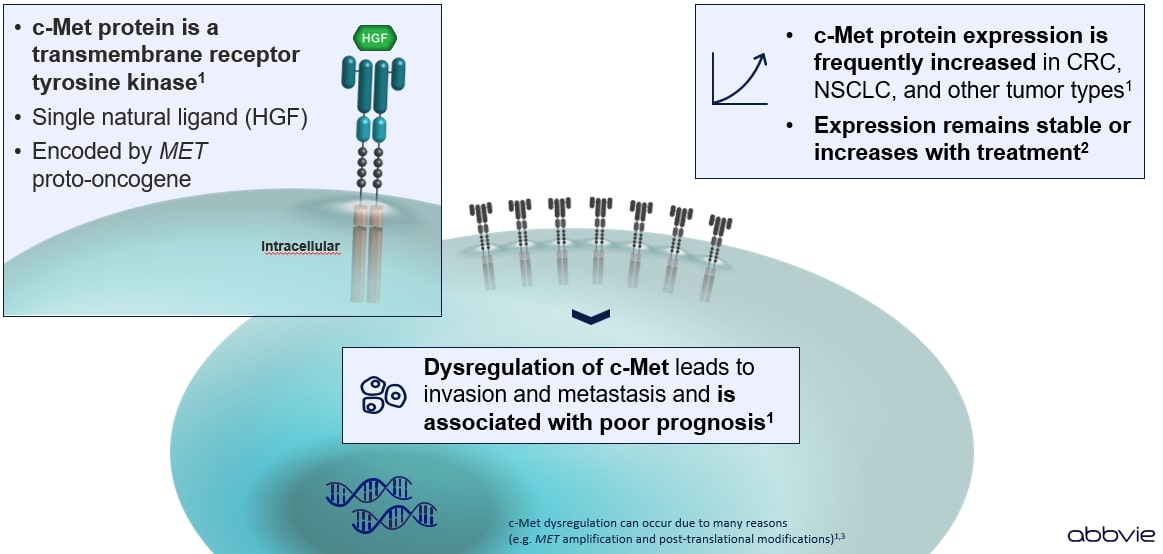
The MET proto-oncogene encodes the c-Met protein (a receptor tyrosine kinase; also known as hepatocyte growth factor receptor, HGFR), which plays an important role in embryogenesis, organogenesis, and wound healing. c-Met has tyrosine kinase activity and is expressed on the surface of epithelial and endothelial cells.1
- The only known ligand for c-Met is hepatocyte growth factor (HGF).1
- When HGF binds to c-Met, it activates multiple intracellular signaling pathways including:1
- The MAPK/RAS cascade
- The PI3K/AKT cascade
- The STAT pathway
- These signaling pathways are responsible for driving proliferation, cell survival, migration and invasiveness.1
- c-Met/HGF are essential for embryonic development and tissue repair.2
IMPLICATIONS IN CANCER
Dysregulated MET signaling in cancer occurs through a variety of genetic, epigenetic, and/or post-transcriptional mechanisms including but not limited to:1-3
- Germline or somatic mutations, chromosomal rearrangement, or MET amplification
- Increased c-Met protein expression
- Alteration of other pathways affecting MET transcriptional activation
- Increased HGF expression
Activated c-Met protein directs intracellular signaling pathways resulting in tumor cell proliferation, cell survival, tumor growth, angiogenesis, migration and invasiveness, resistance to therapy, and maintenance of cancer stem cells.1
Notably, alterations in c-Met and/or HGF appear to confer an increased propensity for a more aggressive clinical behavior manifested by invasion, metastasis, and resistance to the therapy.1
Oncogenic Expression
Depending on the protein expression cutoff applied, increased c-Met protein expression may have prognostic significance in a variety of tumors including:
- Breast cancer 4
- Non-small cell lung cancer5
- Gastric cancer6
- Colorectal cancer 7
- Head and neck cancer8
- Kidney cancer9
- Pancreatic cancer10
Because of the redundant activation of c-Met-directed pathways, c-Met protein expression on tumors by itself is not a sufficient predictor for activity of small molecule inhibitors.11,12
- The antitumor activity of c-Met inhibitors is generally limited to a subset of patients with tumors that are driven predominately by MET signaling.
- Because of this, patient selection is limited to those with MET gene amplification, which is ~2%-5% of the NSCLC population.15
Antibody drug conjugates (ADCs) designed to target c-Met protein represent potential therapeutic strategies that depend on target expression rather than inhibition of downstream signaling for efficacy.11,13
- c-Met protein expression is can be higher in many cancers compared with normal tissues.
Related Research
- Garajová I, Giovannetti E, Biasco G, Peters GJ. c-Met as a target for personalized therapy. Transl Oncogenomics. 2015;7(Suppl 1):13-31.
- Sierra JR, Tsao MS. c-MET as a potential therapeutic target and biomarker in cancer. Ther Adv Med Oncol. 2011;3(1 Suppl):S21-S35.
- Koeppen H, Yu W, Zha J, et al. Biomarker analyses from a placebo-controlled phase II study evaluating erlotinib ± onartuzumab in advanced non-small cell lung cancer: MET expression levels are predictive of patient benefit. Clin Cancer Res. 2014;20(17):4488-4498.
- Lengyel E, et al. C-Met overexpression in node-positive breast cancer identifies patients with poor clinical outcome independent of Her2/neu. Int J Cancer. 2005;113(4):678-82.
- Salgia R. MET in lung cancer: biomarker selection based on scientific rationale. Mol Cancer Ther. 2017;16(4):555-565.
- Mo HN, Liu P. Targeting MET in cancer therapy. Chronic Dis Transl Med. 2017;3(3):148-153.
- Liu Y, et al. Prognostic value of c-Met in colorectal cancer: a meta-analysis. World J Gastroenterol. 2015;21(12):3706–3710.
- Kim JH, et al. Clinicopathological impacts of high c-Met expression in head and neck squamous cell carcinoma: a meta-analysis and review. Oncotarget. 2017;8:113120-113128.
- Macher-Goeppinger S, et al. MET expression and copy number status in clear-cell renal cell carcinoma: prognostic value and potential predictive marker. Oncotarget. 2017;8(1):1046-1057.
- Kim JH, et al. Prognostic value of c-Met overexpression in pancreatic adenocarcinoma: a meta-analysis. Oncotarget. 2017;8(42): 73098-73104.
- Wang J, Anderson MG, Oleksijew A, et al. ABBV-399, a c-Met antibody-drug conjugate that targets both MET-amplified and c-Met-overexpressing tumors, irrespective of MET pathway dependence. Clin Cancer Res. 2017;23(4):992-1000.
- Van der Steen N, Deschoolmeester V, Lardon F, et al. cMET in non-small cell lung cancer: pieces of the puzzle. Cancer Res. 2016;76(14 Suppl):Abstract 2242.
- Strickler JH, et al. First-in-human Phase I, dose-escalation and -expansion study of telisotuzumab vedotin, an antibody–drug conjugate targeting c-Met, in patients with advanced solid tumors. J Clin Oncol. 2018;36:3298-3306.
- Lim EH, Zhang SL, Li JL, et al. Using whole genome amplification (WGA) of low-volume biopsies to assess the prognostic role of EGFR, KRAS, p53, and CMET mutations in advanced-stage non-small cell lung cancer (NSCLC). J Thorac Oncol. 2009;4(1):12-21.
- Liang H, Wang M. MET oncogene in non-small cell lung cancer: Mechanism of MET dysregulation and agents targeting the HGF/c-Met axis. Onco Targets Ther. 2020;13:2491-2510.